I.
Introduction
and summary
A.
Introduction
BAAQMD
application 27215 for authority to construct the Metcalf Energy
Center (MEC) along with CEC
99-AFC-3 is the latest in a series of
applications to build large new gas-fired combined cycle power plants which have come before the California Energy
Commission (CEC) and various California air quality management districts
(AQMDs). Within the last year intervenors CARE and CURE, have pointed out that some of these projects
are quite deficient in their BACT and
other air quality analyses.[1] CARE has participated as an intervenor in
the Delta Energy Center. CARE has filed a complaint with the EPA Office of
Civil Rights over the disparate impacts of air pollutants associated with this
project. With these comments, CVRPCARE has
identified a similar lengthy list of inadequacies in the PDOC for the Metcalf
proposal. CARE expects
that the BAAQMD and the CEC will give serious
attention to these inadequacies. Given the extensive scope of our comments, CARE calls upon
the BAAQMD to re-circulate the PDOC for further comment after responding to
our comments. To go directly to an FDOC and FSA at this stage would deny the public any substantive opportunity to
comment on the many issues, which are
inadequately addressed in this PDOC and the PSA, or not addressed at all.
B.
Summary of CVRPCARE’s
comments
CVRPCARE’s
comments have been grouped into a dozen main areas, which CARE
summarized here. Within each area CARE have attempted to not only identify omissions or
errors in the PDOC (e.g., the absence of a
CO catalyst requirement) but also identify the position, which CARE believe
the BAAQMD should adopt, and the facts supporting that position. CARE have also identified
counter-arguments which the Applicant might make, or has already made elsewhere,
and supplied the District with a response to those arguments.
The key deficiencies in the PDOC and PSA are errors of omission. The PDOC does not contain
a serious top-down BACT analysis
(Section II, below). As a result it
fails to correctly identify BACT for NOx (Section III),
for CO (Section IV), for VOC
(Section VII), and for
startup emissions (Section VIII). Along the
way, the PDOC omits any discussion of other air quality benefits
of catalyst-based CO and NOx control technologies (Section VI) and their lack
of negative PM10 impacts (Section V).
The PDOC and PSA relies on Applicant data, which is either wrong or misleading, leading to
inaccurate conclusions regarding PM10 emissions
and offset requirements (Section IX).
The PDOC and PSA also lacks an adequate analysis of the toxics MEC would emit, emissions whose health
consequences would easily exceed the CEC and BAAQMD thresholds for further
analysis and mitigation (Section X).
Finally, CVRPCARE has
commented on a variety of other matters ranging from the meteorological
modeling in the PDOC to the treatment of the proposed on-site internal
combustion engines to the proposed
condition regarding overlapping startups of the two proposed MEC turbines
(Section XI).
II.
the
pdoc does not include a top-down bact analysis
The project is a major source
whose emissions trigger PSD requirements for NOx and CO. (PDOC, Table 3.) Any major modification subject to PSD must conduct an analysis to
ensure that best available control technology ("BACT") is used. This requirement is set forth in section
165(a)(4) of the federal Clean Air Act, in federal PSD regulations at 40 C.F.R.
§ 52.21(j), and in federal regulations providing the requirements for State
implementation plan (SIP) approval of a State PSD program, at 40 C.F.R. §
51.166(j). For PSD purposes, BACT is
“an emissions limitation… based on the maximum degree of reduction for each
pollutant subject to regulation under [the] Act which would be emitted from any
proposed major stationary source….” (40 CFR § 52.21(b)(12).)
PSD regulations require the
District, as EPA’s delegate, to perform and document an analysis to ensure that
federal BACT is used. (CAA, §
165(a)(4); 40 CFR 52.21(j).) This obligation
is fulfilled by conducting what is known as a “top-down BACT analysis” as
outlined in EPA’s NSR Manual. (NSR
Manual,[2]
Chapter B.) The NSR Manual and the top
down procedure have been accepted by EPA’s Environmental Appeals Board (“EAB”)
“as the most current statement of the Agency’s thinking on BACT issues” and are
routinely used to decide cases involving matters of federal law. (See, e.g.,
Masonite Corporation, 5 E.A.D. 558 (EAB 1994); Inter-Power of New York, Inc.; 5
E.A.D. 135 (EAB 1994); Hawaiian Commercial & Sugar Company, 4 E.A.D. 99
(EAB 1992).)
The Environmental Appeals Board of the EPA has on several occasions stressed the primary importance of a complete and meaningful BACT analysis in the PSD program, stating most recently:
“The BACT
analysis is one of the most critical elements of the PSD permitting process. As
such, it should be well documented in the administrative record. A permitting
authority's decision to eliminate potential control options as a matter of
technical infeasibility, or due to collateral impacts, must be adequately
explained and justified. See In re Masonite Corp., 5 E.A.D. 551, 566 (EAB 1994)
(remanding PSD permit decision in part because BACT determination for one
emission source was based on an incomplete cost-effectiveness analysis); In re
Pennsauken County, N.J., Resource Recovery Facility, 2 E.A.D. 667, 672 (Adm'r
1988) (remanding PSD permit decision because “the applicant's BACT analysis
does not contain the level of detail and analysis necessary to satisfy the
applicant's burden” of showing that a particular control technology is
technically or economically unachievable); Columbia Gulf, 2 E.A.D. at 830
(permit applicant and permit issuer must provide substantiation when rejecting
the most effective technology).” In re
Knauf Fiber Glass, GmbH, PSD Appeal Nos. 98-3 through 98-20 (EAB, Feb. 4,
1999).”
The
top-down BACT process consists of five steps that are discussed in detail in
Section B of the NSR Manual. These
steps are (NSR Manual, Table B-1):
1.
Identify
all control technologies (including lowest achievable emission rate or LAER)
2.
Eliminate
technically infeasible options
3.
Rank
remaining control technologies by control effectiveness
4.
Evaluate
the most effective control and document results
5.
Select BACT
In brief, the top-down process
requires all available control technologies to be ranked in descending order of
effectiveness. The PSD applicant first
examines the most stringent – or “top” – alternative. That alternative is established as BACT unless the applicant
demonstrates, and the permitting authority in its informed judgment agrees,
that technical considerations, or energy, environmental, or economic impacts
justify a conclusion that the most stringent technology is not “achievable” in
that case. (NSR Manual at p. B.2.)
The
PDOC does not contain a responsive BACT analysis.[3] Instead, the PDOC has leapt to step 5,
selecting BACT from its outdated "Guidelines 89.2.1" for gas turbines
(PDOC, pp. 9-10), without conducting the analyses required for any of the
preceding steps. The BAAQMD's guideline
for large gas turbines (>23 MMBtu/hr) was adopted on August 24, 1998, nearly
two years ago. (Exhibit --: Guidelines
89.2.1.[4]) Two new technologies have been introduced
into the marketplace since this guideline was adopted, SCONOx and XONON. Had the District performed a formal top-down
analysis, it would have included these two new technologies which are
technically feasible and achieve lower emissions limits than those proposed for
Metcalf. The District has failed to
identify BACT by failing to perform a top-down BACT analysis.
III.
BACT
HAS NOT BEEN REQUIRED FOR NOx
The PDOC concludes that BACT for
nitrogen oxides ("NOx") is an emission limit of 2.5 ppm at 15% O2
averaged over 1 hour, achieved using dry low NOx combustors
("DLN") and selective catalytic reduction ("SCR") technology,
during all conditions except startups and shutdowns. (PDOC, p. 9.) Ammonia
slip was separately limited to no more than 10 ppm. (PDOC, p. 28, Condition 20(e).)
The Applicant and the District have made this determination without
considering or evaluating two competing technologies, SCONOx and XONON. As described below, BACT for NOx is an
emission limit of no more than 1.3 ppm at 15% O2 averaged over 1
hour and no ammonia slip, which can be achieved by using SCONOx and other
technologies.
A.
SCONOx
Should Have Been Considered In A Top-Down BACT Analysis
SCONOx is a
catalytic system that simultaneously oxidizes carbon monoxide ("CO")
to carbon dioxide ("CO2"), hydrocarbons to CO2
plus water ("H2O"), and nitrogen oxide ("NO") to
nitrogen dioxide ("NO2"). The NO2 is then
absorbed onto a potassium carbonate-impregnated ceramic-based catalyst. Passing a dilute hydrogen reducing gas
across the surface of the catalyst in the absence of oxygen continuously
regenerates the catalyst. The hydrogen
reacts with nitrites and nitrates to form water and elemental nitrogen. Carbon dioxide in the regeneration gas
reacts with potassium salts to form potassium carbonate, which is the absorber
coating that was on the surface of the catalyst before the oxidation/absorption
cycle began.
There is no indication in the
PDOC that any technology other than SCR was considered. This by itself is a clear violation of the
PSD rules. The first step of the
top-down process is to identify all available control options. (NSR Manual, pp. B.5, B.10-B.11, Table
B-1.) SCONOx is clearly among the
technologies available. EPA has stated,
“SCONOx (and possibly in the near future XONON) should be included in a
BACT/LAER analysis for proposed combined cycle gas turbine power plant projects
in Region 9.” (Exhibit --: Haber 3/24/00.[5]) Furthermore, the South Coast Air Quality
Management District (“SCAQMD”) has concluded that SCONOx achieves the Lowest
Achievable Emission Rate (“LAER”) for NOx.
(SCAQMD 5/98: Exhibit 3).
Technologies that satisfy LAER “must also be included as control
alternatives [for BACT purposes] and usually represent the top
alternative.” (NSR Manual, p.
B.5.) Therefore, SCONOx must be
included in the District’s BACT analysis.
A proper top-down analysis would
almost certainly conclude that SCONOx is BACT for this project, even if it
achieved exactly the same emission limit as SCR, because it offers a number of
important advantages over SCR alone, with no offsetting disadvantages. First, SCONOx uses a single catalyst to
simultaneously remove NOx, CO, VOCs, and toxics. Second, it uses no ammonia or other hazardous materials and thus
requires no ammonia slip, eliminating the many significant impacts associated
with ammonia use (e.g., transportation accidents, unloading accidents, site
releases, PM10 generation).
Third, the SCONOx system operates effectively at temperatures ranging
from 300oF to 700oF, making it well suited for merchant
operation and providing better control during startups and shutdowns than achieved
with other competing catalytic technology (e.g., SCR, CO oxidation
catalyst). Fourth, unlike other
catalytic systems, the SCONOx catalyst is continuously regenerated, assuring
continuous maximum catalyst effectiveness.
Finally, notwithstanding the forgoing benefits, SCONOx has achieved much
lower NOx and CO levels than other competing technologies and, therefore, is de facto, BACT for this project. (See Comments I.B.1 and II.B, below.)
There is no indication in the
PDOC that SCONOx was even considered.
The District appears simply to have adopted the Applicant’s choice of
SCR without any scrutiny of any kind, relying only on its outdated
guidelines. The District's guidelines
are no substitute for a formal top-down BACT analysis. Further, if SCONOx was in fact considered
and rejected in favor of a less beneficial technology such as SCR, then the
District must clearly justify its choice in the public view. (See In re Columbia Gulf Transmission Co., 2
E.A.D. 824, 827 (Adm’r 1989) (detailed substantiation required when rejecting
most effective technology).)
The District, as EPA’s delegate,
has an affirmative duty under the Clean Air Act to promote the use of more
environmentally protective technologies.
(74 U.S.C. §§ 7475(a)(4), 7479(3).)
Thus, the District should have either required that the Applicant
perform a proper BACT analysis, or conducted the analysis itself, since a
permit decision must reflect a level of detail and analysis indicating that the
permit issuer has reached a considered judgment. (In re: Knauf Fiber Glass, GmbH, PSD Appeal Nos. 98-3 through
98-20, slip op. at p. 15 (EAB, Feb. 4, 1999); In re Pennsauken Co., N.J.,
Resource Recovery Facility, 2 E.A.D. 662, 667 (Adm’r 1988); In re Ash Grove
Cement Co., RCRA Appeal Nos. 96-4 & 96-5, slip op. at 41 (EAB, Nov. 14,
1997); In re Austin Powder Co., 6 E.A.D. 713, 720 (EAB 1997); In re Gennesee
Power Station, L.P., 4 E.A.D. 832, 835 (EAB 1993); In re Pennsauken Co., N.J.,
Resource Recovery Facility, 2 E.A.D. 667, 672 (Adm’r 1988) (remanding a PSD
permit because the record did not contain the level of detail and analysis
required).)
As discussed below, SCONOx is
BACT for NOx for this project under PSD regulations because it achieves the
"maximum degree of reduction" for NOx and is additionally
commercially available, technically feasible, environmentally superior, and
cost effective. It has significant
environmental advantages compared to its nearest competitor, SCR, because it
eliminates the risks of handling ammonia and degradation of air quality from
the generation of PM10. In the
following sections, CVRP first present the evidence that SCONOx has been
demonstrated to achieve a lower NOx limit than any other technology. CVRP then discuss the evidence that SCONOx
is commercially available, technically feasible, cost-effective, and
environmentally superior.
B.
NOx
Emission Limit Of 2.5 ppmv Is Not BACT
The District concluded that BACT for NOx is an emission limit of 2.5 ppmv @ 15% O2 averaged over 1 hour. Lower NOx levels have been required in permits and achieved in practice. These lower levels should have been included in the District's BACT analysis. The PDOC contains no evidence that the District considered any other emission limit. The NSR Manual requires that "the most effective control option not eliminated in step 4 is selected as BACT." (NSR Manual, p. B.53.) In this section, CVRP demonstrate that a lower NOx limit than proposed by the District, 1.3 ppm, has been achieved in practice. In the next section, CVRP demonstrate that there is no ground for eliminating the technology, SCONOx that has achieved this lower limit.
In 1998, the U.S. EPA concluded,
based on six months of Continuous Emission Monitoring (“CEM”) data, that the
Federal Facility had “demonstrated in practice” NOx emissions rates that are
consistently at or below 2.0 ppmvd based on a 3-hour rolling average. (Haber
3/23/98.[6]) The South Coast Air Quality Management
District (“SCAQMD”) subsequently independently concluded based on this same
data that the Lowest Achievable Emission Rate (“LAER”) for NOx for gas turbines
greater than 3 MW in rated capacity is 2 ppm based on a 3-hour average or 2.5
ppm based on a 1-hour average. (SCAQMD
6/12/98.[7]) LAER emission limits are the top alternative
in a BACT analysis and must be adopted as BACT unless eliminated based on
economic or environmental factors. (NSR
Manual,[8]
p. B.5.) An emission limit of 2.5 ppm
averaged over 1 hour has been adopted as BACT for all large gas turbines
permitted in California since, based on EPA’s and the SCAQMD’s determinations
for the Federal Facility (e.g., Sutter Power Project, High Desert Power
Project, La Paloma Generating Project, Los Medanos Energy Center, Delta Energy
Center).
1.
Federal
CEMs Data Indicates BACT Is 1.3 ppm NOx
The Applicants argue that SCONOx
does not offer any improved performance with respect to NOx control, compared
with SCR (Metcalf Response to CVRPCVRP Data Request 8a) and the
"unappealing characteristics of SCONOx are not offset by any significant
improvement in overall NOx emission reduction performance." However, the most recent CEMs operating data
from the Federal Facility demonstrate that they are wrong. Further, the vendors are willing to
guarantee NOx emission limits of 1 ppm averaged over 1 hour. This is clear evidence of improved
performance compared to SCR.
Since EPA and the SCAQMD made
their BACT determinations based on the Federal Facility in 1998, the
performance of the SCONOx unit at the Federal Facility has been improved by
adding more catalyst[9]
so that it now consistently meets a lower NOx emission limit. Further, it has been converted from a base
load facility to a merchant facility, which means that it is more
representative of the Metcalf Project.
Other parties collected 9 months of CEMs data from the Federal Facility for the period April 1, 1999 through December 31, 1999 and analyzed it to establish the “maximum degree of reduction” or BACT for both NOx and CO.[10] the continuous emission monitors at the Federal Facility are certified annually by the SCAQMD. In addition, Sunlaw conducts annual stratification source tests to verify that the location of the CEMs sampling point in the stack is representative of the average exhaust gas concentration. The results of the stratification test conducted during this 9 month period shows that the CEMs sampling location was within 0.07 ppm or 8.1% of the measured average concentration.
It is important to note that this
data is from the very same facility that was used by both the EPA and the
SCAQMD to establish the 2.5 ppm BACT level that has been widely permitted in
California and accepted by the District for other large combined cycle merchant
plants. Thus, there can be no dispute
that additional data from this same facility is relevant to this Project and is
a reasonable basis to establish a lower BACT NOx level.
The 1-hour average NOx CEMs data
for the modified Federal Facility are summarized in (05-31-00) CVRP comments on
the MEC PDOC, Figure 1. This figure
excludes excursions due to startup, shutdown, and non-SCONOx operating problems
(e.g., CEM failure, plant trips, operator error, condensation in gas
generator), as allowed by the permit.
Rolling averages were calculated from 15-minute data in the file in by
eliminating all valid exceedances based on the operating log and averaging the
remaining data in groups of four 15-minute segments. All such excursions are documented based on the plant-operating
log.
The maximum reported NOx
concentration is 1.275 ppm as a 1-hour rolling average, and 1.254 ppm as a
3-hour rolling average.[11] Figure 1 is based on 9,380 15-minute average
NOx measurements out of a total possible of 25,595 because the plant was
operated in merchant mode and only dispatched about 37% of the time due to mild
weather conditions in 1999. These data
demonstrate that BACT for NOx should be established at 1.3 ppm averaged over 1
hour.
2.
Other
Data Confirm That BACT For NOx Is Lower Than 2.5 ppm Averaged Over 1 Hour
Other
information also confirms that BACT for NOx is lower than 2.5 ppm averaged over
1 hour required for this Project.
First, SCONOx has been installed
on a second power plant, at the Genetics Institute in Andover, Massachusetts,
which is reportedly meeting a 1-ppm NOx limit when the turbine is functioning
properly.
Second, both Massachusetts and
Connecticut have made BACT determinations and issued permits requiring that
large gas turbines achieve a NOx limit of 2 ppmv at 15% O2 averaged
over 1 hour. These permits are based on
clock or block averages, identical to the averaging time proposed for
Metcalf. This is a lower emission limit
than 2.5 ppmv averaged over 1 hour.
Notwithstanding the CEMs data
reviewed above, these permits alone establish a lower NOx emission limit for
Metcalf than that required in the PDOC and therefore should have been
considered by the District. The lowest
permit limit required in any construction permit which has been issued anywhere
in the country in the time period up to and including the public comment period
on a permit establishes BACT.[12] Hence, even if the CEMS data from the
Federal Facility did not establish a new BACT level for this Project (which it
does), the BACT determinations made in Massachusetts and Connecticut, which
specify a NOx limit of 2 ppmv averaged over 1 hour, would constitute a new BACT
level for this Project. Thus, the
District must include 2.0 ppm NOx in its BACT analysis.
3.
NOx
Levels Of 1 ppm Can Be Accurately Measured
In other forums, it has been
argued that the NOx BACT limit should not be lowered because NOx
cannot be accurately measured, even at 2.5 ppm. (CARB 9/99, pp. 24-25.)
These and similar issues were also raised in the South Coast AQMD’s
proceedings in which the 2.5 ppmvd BACT level was originally adopted. The South Coast AQMD thoroughly evaluated
and rejected all of these measurement issues as insignificant in its staff
report (Ex. 7, South Coast AQMD Staff Report) and in correspondence. (Gangule
5/26/98.[13])
NOx levels can be
accurately measured at 1.0 ppm. This
has been demonstrated at the Sunlaw Federal Facility, where three separate
monitoring systems were used there to measure NOx: (1) the facility CEM, which is an API Series
200 CEM with a lower limit of detection and precision of 0.1 ppm; (2) South
Coast AQMD Reference Method 100.1 by South Coast AQMD staff; and (3) in annual
certification tests by an independent source testing firm using the RECLAIM
Rule 2012 protocol.[14] All three testing methods provided
consistent measurement results.
Manufacturers of NOx
CEMs -- Rosemount Analytical, California Analytical Instruments, Thermo
Analytical Instruments, and Advanced Pollution Instruments -- all offer CEMs
that are guaranteed to measure NOx from 0 to 10 ppm with a lower
limit of detection of 0.1 ppmv NOx or 1% of full scale, and a
precision of the greater of either 0.1 ppm or 1% of full scale or better. This is well below the BACT level of 1.3 ppm
CVRP have proposed for TMPP, is consistent with the results of certification
and inter-method comparison studies at the Sunlaw Federal Facility, and
provides a comfortable margin of safety.
Thus, CVRP believe that measurement issues should not limit the
establishment of BACT for gas turbines at 1.3 ppm.
C.
SCONOx
Is Technically Feasible Under PSD Regulations
The
top-down BACT analysis process allows a control option to be eliminated if it
is technically infeasible or results in adverse energy, environmental, or
economic impacts. (NSR Manual, §
B.) The NSR Manual clarifies when a
technology is technically feasible:
“[I] f the control technology has been installed and operated
successfully on the type of source under review, it is demonstrated and it is
technically feasible.” (NSR Manual, p.
B.17.) SCONOx meets this test, as
demonstrated below.
1.
SCONOx
Has Been Installed And Successfully Operated
The SCONOx system has been
demonstrated to achieve 2 ppmvd averaged over 3 hrs or 2.5 ppmvd averaged over
1 hr[15]
on the 32 MW combined cycle (25 MW LM2500 gas turbine plus 7 MW steam turbine)
Federal Cogeneration facility in Vernon, California (“Federal Facility”). (South Coast AQMD Staff Report, p.
3-4.) The South Coast AQMD has
concluded that SCONOx/water injection is “achieved-in-practice” technology for
natural gas-fired turbines with rated capacities of 3 MW or greater. EPA Region 9 has also concluded that the
Federal Facility “has, based on data submitted to EPA for the six-month period
from June 28, 1997 to December 28, 1997, ‘demonstrated in practice’ NOx
emissions rates that are consistently at or below 2.0 ppmvd based on a 3-hour
rolling average.” (Haber 3/23/98.[16]) EPA has recently acknowledged that this same
facility is currently meeting 1 ppm NOx. In addition, SCONOx has been operating
on a 5-MW Solar Taurus 60 gas turbine at the Genetics Institute facility in
Andover, Massachusetts since August 1999, likewise meeting 1 ppm.
Setting aside the more stringent
technical feasibility demonstration discussed above, which SCONOx meets, the
NSR Manual alternatively defines a technology as technically feasible if it is
both “available” and “applicable.” (NSR
Manual, p. B.17.) A technology is
considered “available” “if it has reached the licensing and commercial sales
stage of development.” (Ibid, p.
B.18.)
2.
SCONOx
Is Available
The Applicants have argued that
SCONOx is not BACT because Massachusetts concluded in July 30, 1999, "the
Department cannot conclude that SCONOx is the Best Available Control Technology
for the control of NOx for turbines larger than 50 megawatts." (Metcalf Response to CVRPCVRP Data Request 7a, p. 6.) When this was written, ABB had not completed
it evaluation and scale up program and was not commercially offering
SCONOx. However, ABB Alstom Power
announced on December 1, 1999 that SCONOx is commercially available for large
gas-fired turbines. ABB Alstom Power
has licensed SCONOx from Goal Line and is the exclusive licensee for power
plants larger than 100 MW. Goal Line
itself may sell the SCONOx system if ABB cannot or will not sell SCONOx to a
given applicant. ABB Alstom Power has
completed testing and scale-up of the technology and is now offering it for
sale with performance guarantees, specifically targeting the largest gas
turbines made (e.g., ABB GT-24, Westinghouse 501G) or announced (e.g., GE Frame
7H). The technology is fully described
on ABB's website. (www.apcnoxcontrol.com.)
The ABB announcement was based on the completion of scale-up testing by ABB, which was subsequently reviewed and confirmed by Stone & Webster. This work included a comprehensive review and analysis of design documents for a 270 MW reference plant consisting of one ABB GT-24 turbine, one HRSG, and one steam turbine. A full-scale prototype damper system for this plant was constructed and operated on a test rig for 101,000 cycles, equivalent to about 5 years of continuous operation. Regeneration gas flow distribution through the catalyst was investigated using both computer and physical model studies. A control system failure mode analysis was also performed.
In addition, Marsh USA Inc.
conducted an insurance review and concluded that "CVRP did not discover
any issues that cannot be readily addressed by ABB or which, in our opinion,
make the SCONOx system uninsurable."
They further concluded, "The underwriting community will positively
embrace SCONOx as a viable product that reduces industry emissions. Realistically, due to the method in which
pricing is determined, CVRP expect no additional premium credit being applied
to project insurance if a SCONOx system is installed." (Marsh 10/6/99.[17])
3.
SCONOx
Is Applicable
The NSR
Manual additionally considers a technology to be “applicable” “if it has been
or is soon to be deployed (e.g., is specified in a permit) on the same or a
similar source type.” (Ibid. p. B.18.) The San Joaquin Valley Air Pollution Control
District has specified SCONOx in a permit on a 262-MW ABB GT-24 gas turbine
issued to the La Paloma Generating Co. LLC in an authority to construct permit
issued May 26, 1999,[18]
by the San Joaquin Valley AQMD.[19]) It has also been proposed for use on the
510-MW Otay Mesa Project at 2 ppm with a goal of 1 ppm, and on the 550-MW Nueva
Azalea Project at 1 ppm NOx and 0.5 ppm CO. Therefore, even if SCONOx had not been
installed and successfully operated on two gas turbines representing the same
“type” of source, it would still be judged to be technically feasible using
EPA’s alternate criterion for situations where there is no actual operating
experience.
4.
EPA
Has Concluded SCONOx Is Technically Feasible
Finally, the U.S. EPA has
determined that SCONOx is technically feasible. EPA Region 1 recently concluded, based on correspondence with ABB
Alstom Power, that “it is our view that SCONOx is a technically feasible
control option for large combined cycle turbine project” and “the Region now
considers SCONOx a technically feasible and commercially available air
pollution control technology that is expected to obtain emission levels for
criteria pollutants such as NOx, CO and VOC comparable or superior
to previously applied technologies for large combined cycle turbine
applications.”[20] The EPA and ABB correspondence supporting
this conclusion are included in (5-31-00 CVRP comments on MEC PDOC, Ex. 16:
Region I letter).
5.
There
Is No Demonstration Of Technical Infeasibility
The NSR Manual notes “the control option is presumed to be
technically feasible unless the source can present information to the
contrary.” (NSR Manual, p. B.19.) The demonstration of technical infeasibility
must be “based on a technical assessment considering physical, chemical and
engineering principles, and/or empirical data showing that the technology would
not work on the emission unit under review, or that unresolvable technical
difficulties would preclude the successful deployment of the technique.” (Id., p. B.20.) No such demonstration has been made in this case. However, the Applicants have argued in other
fora that SCONOx is not technically feasible.
In material handed out in an
Energy Commission air quality workshop
on MEC,
held in San Jose on April 5, 2000 ---, the Applicant argues that SCONOx
"is not feasible for the following reasons: 1) significant adverse impacts
on plant reliability, 2) significant degradation of plant performance and
output, 3) increase in the complexity of instrumentation and controls, 4) significant
increases to plant capital and operating costs, 5) the reality that the system
has never been proven, much less even test, on large gas turbine, and 6)
satisfactory commercial performance guarantees are unlikely."[21] All of these claims are false. The vendor, ABB, reviewed these claims and
prepared a response, dismissing each allegation. (Oegema 4/14/00.[22])
In responses to data requests
propounded by CVRPCVRP,
the Applicants additionally argue that: 1) SCONOx is not commercially available
for Otay Mesa and at any rate would not achieve lower emission limits; 2) that
the Nueva Azalea proposal is irrelevant because Sunlaw, the proponent of Nueva
Azalea, has a financial stake in SCONOx; 3) that SCONOx triples PM10 emissions;
and 4) that Massachusetts has concluded that SCONOx is not BACT in certain
cases, among others. (Metcalf Responses
to CVRPCVRP
Data Requests 6-9.[23]) Many of these allegations are based on out
of data information. All of them are
incorrect.
The following subsections discuss
each allegation made by MetcalfCalpine/Bechtel Enterprises.
a. SCONOx Can Be Scaled
Up
The Applicant argued that SCONOx
"has never been proven, much less even tested, on large gas turbines"
and "SCONOx technology to date has only been tested on two small gas
turbines...more catalyst modules of the same size used at the test plant would
be required if this technology is applied to a large industrial frame
combustion gas turbine. Calpine/Bechtel
have serious concerns about this unproven system and its impact on reliability,
and more importantly, its ability to maintain compliance with all applicable
emissions limits." This is
incorrect.
The definition of BACT only
requires that the technology be “achievable for such source or modification
through application of production processes or available methods, systems, and
techniques . . ..” (40 CFR § 52.21(j)
(emphasis added).) Restricting the
application of new technologies to the exact type and size of unit initially
tested would unnecessarily limit the transfer of technology and defeat the
purpose of the Clean Air Act. In fact,
the case-by-case approach selected by Congress to implement BACT was
specifically designed to cause “the adoption of improvements in technology to
become widespread far more rapidly than would occur with a uniform Federal
standard.”[24] The arguments advanced by the Applicants
turns this goal on its head.
Nevertheless, there is no
evidence that size and type of turbine affects the performance of SCONOx. EPA has unequivocally stated that SCONOx is
technically feasible for large combined cycle projects such as this one. EPA Region I recently concluded, based on
correspondence with ABB Alstom Power, that “it is our view that SCONOx is a
technically feasible control option for large combined cycle turbine project”
and “the Region now considers SCONOx a technically feasible and commercially
available air pollution control technology that is expected to obtain emission
levels for criteria pollutants such as NOx, CO and VOC comparable or superior
to previously applied technologies for large combined cycle turbine
applications.”[25]
In addition, both EPA and other
regulatory agencies that have considered scale-up of SCONOx to larger size
turbines have concluded that scale-up is not a concern. As far back as 1998, EPA Region 9 concluded
there are no scale-up issues. “In June
1998, Region 9 informed the South Coast Air Quality Management District (AQMD)
that CVRP were not aware of any technical problems associated with scale-up of
the SCONOx technology to larger turbines.” In 1999, EPA Region 1 reiterated,
“there are no known scale-up concerns with SCONOx. Consequently, it is our view that SCONOx is a technically
feasible control option for large combined cycle turbine projects.”
The SCAQMD rigorously evaluated the scale-up issue in its BACT/LAER determination and concluded that:
It is the
staff’s technical finding that the SCONOx control technology can be scaled up
in comparison to the 32 MW demonstration plant since the exhaust
characteristics of the turbines are similar.
Based on staff review of AQMD source test reports for different
turbines, staff finds that the NOx reduction process and the characteristics of
the exhaust gases from natural gas fired turbines are similar regardless of
size above 3 MW. Therefore, the
identified emission rate of 2.5 ppm NOx at 1-hour average constitute BACT/LAER
for gas turbines with rated capacities of 3 MW or larger. U.S. EPA staff also has the same technical
judgment concerning this issue. (BACT
Guidelines Update 6/12/98, p. 4.)
This position is echoed
throughout the documentation supporting the SCAQMD’s BACT/LAER determination
that is currently used throughout California, including statements that:
“[t]here is no known technical limitation that would render the exhaust flue
gas of a large industrial turbine to have different characteristics than
exhaust from a 30 MW aero derivative turbines” (id., p. 3-4); and “[s]ince
there is no known technical reason that will render the exhaust flue gas from a
large gas-fired turbine to have different characteristics than exhaust from a
30 MW turbine, AQMD staff has concluded that LAER, as presented in the Staff
Report, must apply to gas turbines over 3 MW size.”
This position makes sense because
catalyst systems are designed based on desired removal efficiency, space
velocity, exhaust gas flow rate, and operating temperature. The basic principles of catalyst design
apply regardless of the specific type of catalyst or the size of the catalyst
system. (Heck and Farrauto 1995.[26]) The size of the system only affects the size
and number of components (e.g., dampers, module), the volume of catalyst, and
amount of chemicals, not the fundamental design. The operating temperature in the HRSG where a SCONOx system would
be installed is identical for Metcalf's Westinghouse 501F turbines and the
Solar Taurus on which SCONOx has been demonstrated. For identical temperatures, the amount of catalyst is directly
proportional to the amount of exhaust gas.
Therefore, the amount of catalyst would be greater for a Westinghouse
501F than for the Taurus on which SCONOx has been demonstrated. However, the engineering principles that
apply are the same, regardless of the scale of the installation.
Further, SCONOx and other
catalyst systems, including SCR, are designed in standard module sizes of
similar geometry allowing for unlimited scale-up. This is accomplished by putting standard modules together as
building blocks to obtain a desired configuration.
Successful scale-up has already
been demonstrated for SCONOx, which has more than 10 years of operating
history, much more than many of the new turbines being put into service. SCONOx was originally scaled up from a
laboratory “plug” catalyst the size of a small carrot to slip-stream testing
the size of two cement bricks, to full-scale testing 100 times bigger than the
slip-stream catalyst, to commercial operation at the Federal Facility, to
scale-up testing to >100 MW by ABB, who has licensed the technology for gas
turbines rated 100 MW and larger.
(Stone & Webster 2/22/00.)
Otay Mesa, a project similar to
Metcalf located in the San Diego area,
has proposed to use SCONOx. The
Applicants suggest that as of January 5, 2000, Otay Mesa was uncertain whether
SCONOx was commercially available.
(Metcalf Response to CVRPCVRP
Data Request 6d.) However, much has
changed since then. Otay Mesa is
currently permitting SCONOx. Bob
Hilton, Vice President, and Rick Oegema, SCONOx Product Manager, for ABB
appeared at the March 2, 2000 Otay Mesa status conference before the Energy
Commission and presented testimony on the status of SCONOx. The transcript
reveals that ABB is the largest vendor of pollution control systems in the
world (3/2/00 RT 23:3-7) and is “extremely good” at scaling up technologies and
developing them into full commercial application. (3/2/00 RT 23:15-18.) Mr.
Hilton testified that “CVRP know the system works. It’s been in operation.
We’ve gone through a scale-up program.
And to verify our scaled-up design we’ve actually gone through a
secondary verification program.”
(3/2/00 RT 28:21-24.) The
scale-up of SCONOx “is a routine process with virtually every technology we’ve
taken to market for the last 40 years... And what you’re seeing is not a new
process. This is a developmental
process that we’ve done for years where we’ve just literally scaled things up
in roughly the same order of magnitude that we’re talking here.” (3/2/00 RT 29:6-14.) Mr. Hilton concluded that the scale-up
program that ABB has completed for SCONOx “has basically satisfied us.” (3/2/00 RT 39:20-21.)
Scale-up is simply not an issue
for monolithic modular catalyst systems, such as SCONOx and SCR. There is nothing anywhere in the technical
literature to the contrary. No party
has provided any evidence to the contrary.
Further, regulatory agencies that have reviewed the issue have concluded
there is no scale up issue.
b. Dampers Are Reliable
The Applicant argues that "[t]he SCONOx process uses a set of dampers to isolate the regeneration process from exhaust gases. The power industry normally avoids the installation of any moving parts in the exhaust stream of combustion devices due to the high potential for warping, jamming, corrosion, and ultimate failure of the devices." This is simply not true and is refuted by actual operating experience, scale-up testing performed by ABB, the vendor's comments, and actual experience in the power and other industries.
The SCONOx system is shown
schematically in the figures on the vendor's website (www.apcnoxcontrol.com). The catalyst system consists of 10 to 15
vertically stacked rows of catalyst.
Pollutant removal and catalyst regeneration occur in cycles. Dampers isolate the catalyst rows so that
20% of the rows are being regenerated while 80% are actively removing
pollutants. These dampers open and
close once every 15 to 40 minutes on a continuous basis between major
shutdowns. (Id., p. 4-12.)
SCONOx dampers have been in
continuous operation on the Federal Facility since 1996 and on the Genetics
Facility since July 1999. These dampers
have not caused plant shutdowns.
Maintenance has been minimal, performed with the plant on line. In addition, Mader Dampers, a Division of
Mader Machine Company Inc. of LaGrange, Ohio built a full-scale test rig to
verify the performance and reliability of the damper louvers, seals, seal
design and actuators. Complete,
full-size dampers (30 ft x 3 ft 7-1/2 in with a total seal length of 292 feet),
including seals and actuators, were tested at normal HRSG operating conditions. The test rig was maintained at 610 F by
circulating hot flue gas from an external combustor. No attempt was made to simulate the flow or emissions as these
were evaluated and deemed to have no impact on the test as designed. The dampers were cycled at an accelerated
rate through 101,000 cycles to simulate five years of continuous operation in
four months of testing, representing more severe service than would be
experienced in practice.
These tests demonstrated that the
system performed without much problem up to 60,000 cycles, which is equivalent
to about 3 years of continuous operation.
The typical failure rate of wearable components throughout the power
island is 1 to 3 years. Thus, this is
typical performance, consistent with industry-wide standards.
The only damper issues identified
after 3-years of continuous operation were with the actuators and shaft-seal
interface. The limit switches within the actuators control the damper
sequencing. The original Auma limit
switches were inadequate for the proposed service and thus replaced by external
mechanically actuated limit switches, which resolved the problem. Regardless, these switches costs about $500
and can be replaced in about half an hour without taking the system
offline. Similarly the shaft design was
modified to eliminate shaft-seal leakage. Thus, the modified damper system
currently offered by ABB and Goal Line has none of the problems identified
after 5 years of continuous operation on the test rig. The vendor concluded that “no known
maintenance issues have been identified as part of this design verification”
and “the expected maintenance...can be reasonably performed with typical plant
outage schedules required to service boiler systems and the turbine and should
not impact reliability”, consistent with actual operating experience at the
Federal Facility. The dampers
themselves are designed for 30 years of operation, subject to regular
maintenance, inspection and repair, excluding wear components such as bearings,
seals, actuators, or similar wearable components.[27]
There is simply no evidence that
the dampers would cause reliability, maintenance, and scale-up problems as
alleged by the Applicants. Similar
dampers are widely used throughout the power industry in air pollution control
applications with minimal operational problems as well as in other industries
under conditions far more severe than those that would be experienced by the
SCONOx dampers. (Makansi 1993.[28]) They are also used in bypass applications in
the power industry, when more than one turbine, stack, or other equipment is
present, and are used to isolate equipment from exhaust gases to allow man-safe
access to perform maintenance while the balance of plant remains in operation. In fact, the original SCONOx damper design
was scaled down from large coal plant designs.
Dampers are also widely used in the steel industry in applications that
involve higher temperatures, more corrosive environments, and similar duty
cycles. The dampers used in the SCONOx
process have the added benefit, compared to these other applications, of
cycling full stroke at regular intervals, open to close. The full stroke ensures even wear, and the
regular interval of actuation prevent binding and corrosion due to extended
periods of inactivity that occur in some other applications.
c. The Control System Is
Reliable
The dampers are operated by an
electronic control system. The
Applicants allege that SCONOx "will require a significant addition to the
plant control system and field instrumentation." This is incorrect. The
control system has been in operation at the Federal Facility since 1996 and at
the Genetics Facility since July 1999, and no problems have been
encountered. The vendors provide the
SCONOx instrumentation and control system as a package with a dedicated PLC
controller. This can be simply
connected to the existing plant distributed control system through “PC Highway”
connections. The vendor for their
ruggedness and known reliability selected Field instrumentation and supporting
devices used in this system.
d. Catalyst Regeneration
Will Not Cause An Explosion Hazard
Passing a
controlled mixture of regeneration gases across the surface of the catalyst in
the absence of oxygen continuously regenerates the SCONOx catalyst. The regeneration gases consist of steam with
small amounts (2%-4%) hydrogen and natural gas, depending upon system
design. The hydrogen is required to
convert nitrites and nitrates to water and inert nitrogen gas, not to avoid an
"explosion caused by hydrogen inside the SCONOx modules" as alleged
by the Applicants. The vendor has
corrected most of the Applicants' incorrect statements about the hydrogen
regeneration process.
There is
simply no explosion hazard. Goal Line
conducted a safety evaluation of hydrogen use in the SCONOx process and
concluded that "The use of hydrogen as a regeneration gas does not result
in an unsafe operating conditions for unlikely but possible anomalies that
could result during the operation of the SCONOx system."[29]
The SCONOx system is designed to
prevent explosions. Hydrogen (and
natural gas) is only explosive in the presence of oxygen and an ignition source
and then only if its concentration exceeds the lower flammable limit ("LFL"),
which is 4%. (NFPA 1991, p. 325M-59.[30])
The concentration of hydrogen in the
regeneration gas is typically 2%, half of the LEL. Therefore, even if oxygen and an ignition source was present,
which they are not, or even if hydrogen leaked out of the catalyst modules, the
concentration of hydrogen is too low to cause an explosion.
The hydrogen is only used for
regeneration, which occurs in an inert atmosphere. Oxygen is absent in an inert atmosphere. The purpose of the
dampers that isolate the catalyst modules, discussed above in Section III.C.5,
is to isolate the section of catalyst being regenerated, specifically to
exclude oxygen. Therefore, explosions
are not chemically or physically feasible within the modules.
Even if the right concentration of hydrogen and oxygen were present, an explosion still would only occur if an ignition source were present. There is none for SCONOx because the hydrogen is isolated from the duct burners by the dampers, which seal off the area where the hydrogen is used. Even if leakage occurred, the concentration of hydrogen (and natural gas, depending upon system design) is far below the LFL of 4% for hydrogen and natural gas. The total regeneration gas volume represents only 1% of the turbine exhaust gas volume. Therefore, under worst-case conditions, if leakage occurred, the hydrogen and natural gas concentrations in the flue gas outside of the SCONOx modules, would be no more than 0.04%, which is substantially less than the LFL of 4%. Therefore, this does not pose any flammability or explosion hazards.
In fact, anhydrous ammonia, which
is used in much larger amounts by the proposed SCR installation, also forms
explosive mixtures with air and can ignite in the presence of a combustion
source. (NFPA 1991, p. 325M-14.) The explosion risk is similarly managed by
keeping the maximum concentration of ammonia below the LFL. However, the explosion danger is far smaller
for SCONOx than for SCR because in the SCONOx system, the hydrogen is isolated
from the ignition source and is used in an inert atmosphere with no oxygen,
unlike the ammonia in an SCR system. The Applicant has not expressed any
concerns about the explosion hazards of the aqueous ammonia system required by
its SCR.
The Applicant also asserts that
the reforming process used to generate hydrogen "has never been
successfully tested." This is not
true. There are two hydrogen production
processes that are used with SCONOx.
The first has been in operation since December 1996 at the Federal
Facility. The second has been in
operation since July 1999 at the Genetics Institute Facility. Further, the steam reforming process used in
both of these processes has been widely used throughout the refining,
petrochemical industries, and other industries for decades[31].
Finally, it is important to note
that hydrogen is already widely used in the power industry to purge the
generators. Hydrogen use statistics
have not been reported for Metcalf.
However, other similar facilities typically store about 175,000 cubic
feet of compressed hydrogen on site and another 55,000 cubic feet inside
generators and distribution piping.
(e.g., see Elk Hills AFC, p. 5.12-2.)
The Applicants have not expressed any concerns about either leakage or
explosion hazards of this hydrogen.
e. SCONOx Does Not
Increase Back Pressure
The Applicants allege that the
"pressure drop across the SCONOx catalyst is twice that of a conventional
SCR." SCONOx simultaneously
removes both CO and NOx and is therefore equivalent to a
conventional SCR and oxidation catalyst.
Although the PDOC does not recommend an oxidation catalyst as BACT for
CO and VOCs, an oxidation catalyst is clearly BACT for Metacalf, as discussed below in Comment
--. Further, the Energy Commission has
recommended an oxidation catalyst in the Preliminary Staff Assessment. (PSA, p. 41.)
Vendor quotes indicate that the
backpressure created by SCONOx is about the same to slightly lower than that
created by an SCR and oxidation catalyst combo. The backpressure for SCONOx quoted by ABB for similar applications
is 4.0 inches of water. A similarly
sized SCR would create 3.5 inches and an oxidation catalyst would create 0.8
inches of backpressure. Therefore, the
total backpressure for the conventional system, 4.3 inches of water, is greater
than the backpressure created by SCONOx, the reverse of what is alleged. Further, broadening the area of the catalyst
bed to reduce the backpressure can configure the housing configuration for
SCONOx, as well as other catalytic systems.
Therefore, SCONOx does not cause a 1.2 MW reduction in electrical output due to changed backpressure, as alleged by
the Applicant.
f. SCONOx Steam
Consumption Is Minor
The Applicants allege that 40,000 lb/hr of superheated steam must be extracted from the supply to the steam turbine, which would be lost out of the HRSG stacks, reducing output by about 4.5 MW. This is incorrect.
The regeneration of the SCONOx catalyst requires about 20,000 lb/hr of 600 F, low-pressure steam, which is taken from the steam turbine exhaust where the impact of steam demand on turbine operations would be minimal. GT PRO simulations indicate that this would reduce output by 1 Mw, a minimal amount on a 600 Mw project
-------. Further,
this amount of steam is minor, comprising less than 1% of turbine exhaust
steam. The SCONOx system uses a steam
condensing system that captures 75% to 80% of the regeneration gas steam and
returns it to the boiler condensate and make-up water system to reduce the
make-up water demand. This increases
water demand by about 12,000 gpd per turbine.
g. Commercial Warranties
The Applicant "believes that
the SCONOx system is not commercially available for the turbines proposed at
MEC with commercial performance guarantees adequate to ensure safe and reliable
operation, and continuous compliance with all applicable emission limits, at
the present time." (Metcalf
Response to CVRPCVRP
Data Request 8.) No evidence is
presented to support this position, which is pure speculation.
The vendors are actively quoting
and providing commercial guarantees for SCONOx. ABB is currently working with two serious purchasers for large
Frame 7 engines -- Nueva Azalea and Otay Mesa.
The guarantees currently being offered by ABB are consistent with the
NOx and CO limits that have been demonstrated at the Federal and Genetics
Facilities.
Nueva Azalea has been guaranteed
by ABB and is currently being permitted at 1 ppm NOx and 0.5 ppm CO averaged
over 1 hour. The Applicants argue that
this is irrelevant because "the Nueva Azalea project is being developed by
Sunlaw Energy, which has a financial stake in the marketing of the SCONOx
system. For these reasons, we do not
find the Nueva Azalea Project to be similar to MEC for purposes of determining
the commercial availability of the SCONOx system." (Metcalf Response to CVRPCVRP Data Request 6d.) However, ABB has stated that it will offer
the same guarantee and terms to other applicants, including Metcalf.
The Applicants also argue that
Otay Mesa is not relevant because "in the case of the Otay Mesa Generating
Project, identical emission limits for all pollutants are proposed for all
criteria pollutants regardless of whether SCONOx is used." (Metcalf Response to CVRPCVRP Data Request 8a.) This is not true. The Otay Mesa Project will achieve lower emission limits than
those currently proposed for Metcalf.
The Project is being permitted at 2 ppm NOx averaged over
three hours with a goal of 1 ppm NOx (3/2/00 RT 19:10-12, 46:16-18)
and 100 ton/yr of NOx.
(3/2/00 RT 43:19-20.) The 100
ton/yr limit would require year-round operation below 2 ppm. (3/2/00 RT
46:23-25, 47:1-3, 48:12-17.) Recent
conversations with PG&E Generating indicate that 100 ton/yr actually
corresponds to a concentration limit between 1 ppm and less than 2 ppm NOx,
depending upon the number of startups and shutdowns and actual hours of
operation. Further, the 100 ton/yr
limit proposed for Otay Mesa is substantially lower than the 185 ton/yr
proposed for Metcalf. (PDOC, Condition
25.)
A guarantee is an opening offer,
i.e., ABB’s opening position in what would ordinarily be a negotiation between
a serious buyer and seller. ABB’s
standard warranty for SCONOx is 1 year for equipment and 3 years for catalyst,
which is consistent with industry standards for SCR and other catalytic
systems. A 10-year warranty is also
available, contingent upon the owner entering into a long-term service
agreement for maintenance of the
catalyst.
h. Financing
The Applicants argue that
"financial institutions will be reluctant to provide financing to the MEC
project utilizing SCONOx. Proving project
viability to these institutions would be difficult, if not insurmountable
task. Performance guarantees supplied
by the EPC contractor would be another likely point of contention.." This is demonstratably false.
The
Genetics Facility in Andover, Massachusetts, and the Federal Facility in
Vernon, California both include SCONOx and both have been financed and are
operating. Further, the La Paloma
Project was permitted and financed with SCONOx and is currently under
construction.
Both Otay Mesa, proposed by
PG&E Generating, and Nueva Azalea, proposed by Sunlaw, has been proposed
with SCONOx. Obviously, both project
proponents are confident they will obtain financing or they would not have
proposed SCONOx. Although PG&E
Generating decided not to build La Paloma with SCONOx due to a timing issue
(see footnote --), clearly, SCONOx posed no financial or other obstacle as
PG&E Generating has again proposed to use SCONOx on a second project, Otay
Mesa.
i. SCONOx Is Cost Effective
The Applicants argue that the
installed capital cost of SCONOx is three to four times higher and the
operating costs two to three times higher than a conventional SCR system. This is irrelevant and incorrect.
First, it is simply irrelevant
whether one technology which achieves much higher emission reductions (SCONOx)
costs more than another with lower emission reductions. The key question is whether the more
expensive technology is cost-effective, regardless of how much more it may cost
than a cheaper and less effective technology.
Cost effectiveness is an economic criterion used to assess the potential
for achieving a control objective at the lowest cost. Cost is measured in terms of annualized control costs, and
effectiveness is measured in terms of tons of pollutants removed per year. Therefore, cost effectiveness is the total
annualized costs of control divided by annual emission reductions. (NSR Manual, p. B.36.) The EPA has developed guidelines for
determining cost-effectiveness (NSR Manual, § IV.D.2.b) and procedures to
perform the requisite calculations.
(OAQPS Manual.[32])
Second,
vendor cost data indicates that while installed capital costs for SCONOx are
about twice as high as an SCR/oxidation catalyst, the annual operating costs
for SCONOx are lower than the conventional system. The overall cost effectiveness of SCONOx well within BAAQMD and
other agency guidelines.
Specifically, recent independent cost-effectiveness analyses based
on EPA guidelines and procedures indicate that SCONOx costs $6,270 per ton of
NOx (“$/ton”) to reduce NOx from 25 ppm to 2 ppm on a GE Frame 7
machine. (OnSite Sycom 10/15/99, Table
A-7.[33]) Cost estimates prepared by CURE for Elk
Hills indicate that SCONOx costs $7,360/ton to reduce NOx from 9 ppm to 2.5 ppm
on a GE Frame 7 machine.[34]
We prepared a cost effectiveness analysis for the Metcalf Project that assumes BACT emission limits of 2.5 ppm NOx and 6 ppm CO. Our calculations, summarized in Table 1, are based on vendor-supplied costs and emissions from the PDOC, assuming inlet concentrations of 25 ppm NOx and 10-ppm CO. (PDOC, pp. A-2 to A-3.) Our calculations give no credit for emission reductions during startup, and thus overstate the cost per ton of emissions reductions using SCONOx. Nevertheless, these calculations indicate that SCONOx costs between $5,651 and $6,028 per ton of NOx removed. The cost effectiveness drops to $4,601 to $5,440 per ton when both nonattainment pollutants are included in the calculation. This is reasonable here and has been requested by EPA for other facilities[35] because SCONOx controls both pollutants simultaneously.

All of the SCONOx costs discussed above, ranging
from $4,601per ton of CO or NOx to $7,360 per ton of NOx alone, are well within the BAAQMD’s cost-effectiveness
criterion.
These costs are well within cost-effectiveness criteria
established by major
air districts throughout California. In
the SJVUAPCD, the maximum cost per ton of NOx controlled that is considered
cost effective is $9,700/ton.[36] In the Bay Area Air Quality Management
District, the maximum cost per ton of NOx controlled that is
considered cost-effective is $17,500. [37]
In the South Coast Air Quality Management District, the maximum cost per
ton of NOx controlled that was formerly
considered cost-effective is $17,000/ton.
[38]
The South Coast no longer uses cost-effectiveness criteria.[39]
(Exhibit 6.) Therefore,
SCONOx is cost-effective.
D. OTHER
the BENEFITS
OF SCONOx NEED TO BE CONSIDERED IN DETERMINING
BACT
The SCR system proposed for use
by the Applicants results in a number of environmental problems that are
reduced or eliminated with the use of SCONOx.
These problems include: (1) hazards from accidental releases of the
ammonia used in the SCR system during its transportation and handling; (2) the
formation of particulate matter from the oxidation of SO2 in the SCR
catalyst; (3) the formation of particulate matter from reactions between
ammonia and SO2; (4)
generation and disposal of the hazardous SCR catalyst at the end of its useful
life; (5) inability to control NOx and CO emissions during startups and
shutdowns; (6) increase in NO2 from the use of dry low NOx
combustor.
SCONOx and SCR are compared in
Table 2. SCONOx offers a number of
important advantages over a conventional SCR, with no known disadvantages. First, SCONOx uses a single catalyst to
simultaneously remove NOx, CO, and VOCs.
A conventional system requires two separate catalyst systems. Second, SCONOx uses no ammonia or other
hazardous materials and thus requires no ammonia slip, eliminating the many
significant impacts associated with ammonia use (e.g., transportation
accidents, unloading accidents, accidental releases of ammonia, PM10
generation). Third, the SCONOx system
operates effectively at temperatures ranging from 300oF to 700oF,
making it well suited for merchant power plant operation and providing better
control during startups, shutdowns, and load swings than achieved with other
competing catalytic technology. Fourth,
unlike other catalytic systems, the SCONOx catalyst is continuously
regenerated, assuring continuous maximum catalyst effectiveness. The performance of a conventional catalyst
system, on the other hand, degrades with use.
Fifth, SCONOx has been demonstrated to
achieve much lower NOx and CO levels than other competing
technologies, at lower overall costs.
Finally, the SCONOx catalyst contains precious metals, which can be
reclaimed by smelting, reducing overall operating costs. The SCR catalyst must be disposed as a
hazardous waste at the end of its useful life.
The ammonia impacts and startup/shutdown emissions are discussed further
below.
1. SCONOx Eliminates Ammonia Impacts
The District selected SCR with an ammonia slip of 10 ppm as BACT for NOx without performing a proper environmental analysis as required by the top-down BACT process. (See NSR Manual at B.6.) PM10 is formed from two sources in an SCR system -- ammonia slip and oxidation of SO2 to sulfur trioxide (SO3) by the SCR catalyst. Ammonia and sulfur trioxide form particulate matter within the stack and secondary PM10 downwind in the atmosphere. This PM10 can cause health impacts, visibility impairment impacts, contribute to existing exceedances of the California ambient PM10 standard, and impact listed and protected species. These collateral increases in PM10 have not been evaluated. Further, ammonia is a hazardous substance, and the transportation and handling of ammonia can result in accidental releases that adversely impact the public.
2. Ammonia Impacts Must Be Considered
The top-down BACT process and the federal law it implements require that the environmental impacts of selected technologies be considered as part of the BACT determination. The federal Clean Air Act defines BACT as an “emission limitation” that is set “on a case-by-case basis . . . taking into account energy, environmental, and economic impacts and other costs.” (42 USC § 7479(3), (emphasis added).) The Environmental Appeals Board has provided the following interpretation of the emphasized portion:
[I]f application of a control
system results directly in the release (or removal) of pollutants that are not
currently regulated under the Act, the net environmental impact of such
emissions is eligible for consideration in making the BACT determination. [As a result of the analysis], the control
system proposed as BACT may have to be modified or be rejected in favor of
another system. In other words, EPA may
ultimately choose more stringent emission limitations for a regulated pollutant
than it would otherwise have chosen if setting such limitations would have the
incidental benefit of restricting a hazardous but, as yet, unregulated
pollutant. (North County Resource
Recovery Associates, 2 E.A.D. 230 (EAB 1986).)
The U.S. EPA has similarly
interpreted this requirement to mean that, where two technology choices provide
equivalent control for a regulated pollutant, but one would also control
pollutants not directly regulated by the PSD Program, such as PM10 and NH3
in this case, the one controlling the unregulated pollutants should be chosen
as BACT.[40] Moreover, EPA is seeking to integrate
pollution prevention as an ethic throughout its activities. (Habicht 5/28/92.[41])
The NSR Manual itself is clear
that the environmental analysis should also include a consideration of
“...visibility impacts, or emissions of unregulated pollutants.” (NSR Manual, p. B.46.) Thus, even if SCONOx did not achieve lower
NOx limits than other technologies, SCONOx should have been deemed
BACT on the basis that it eliminates ammonia emissions.
6.
a. PM10 Formation From Oxidation
Of SO2 And Ammonia Slip
The SCR catalyst oxidizes SO2 to SO3. The excess residual ammonia downstream of the SCR system (i.e., the slip) reacts with this SO3 as well as NO2 and water vapor in the stack gases and downwind in the atmosphere to form ammonium sulfate, ammonium bisulfate, and ammonium nitrate according to the following reactions. (Seinfeld and Pandis 1998, pp. 529-534;[42] South Coast AQMD 6/12/98, p. 3-3; Matsuda et al. 1982;[43] Burke and Johnson 1982.[44])
SO3 + 2 NH3 D (NH4)2SO4 (1)
SO3 + NH3 D NH4HSO4 (2)
NO2 + OH + NH3
D
NH4NO3 (3)
The resulting salts
form particulate matter, which contributes to ambient PM10, causes maintenance
problems in the HRSG, contributes to visibility impairment, and impacts
protected species. These salts cause a
number of environmental problems, which are addressed below.
i. PM10
Formation Contributes To Existing Violations Of State Standards
The reactions between SO3, NH3, and NO2 form salts, some of which are emitted to the atmosphere and some of which deposit within the HRSG. The above equations can be used to estimate a portion of the secondary PM10 that is formed from ammonia slip. Secondary PM10 can be formed by reaction of ammonia with SO3 and NO2 emitted by the gas turbines and present in the stack gases and plume as well as additional SO3 and NO2 that are present downwind in the atmosphere.
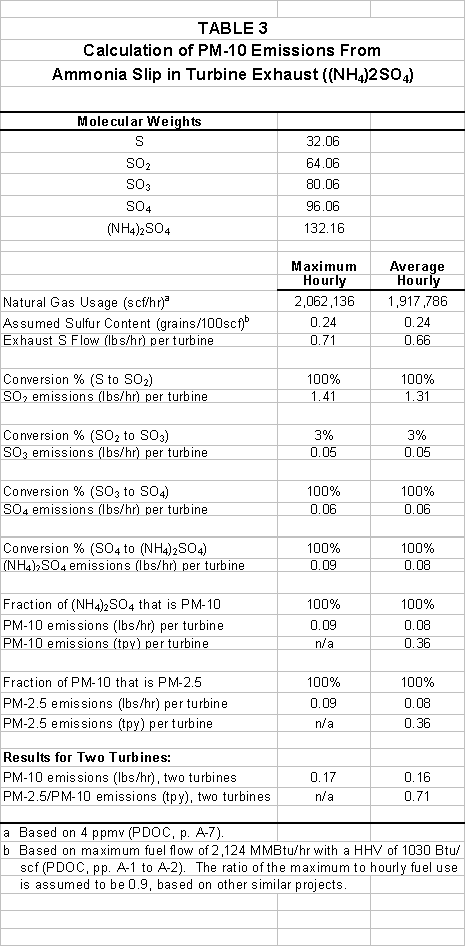
CVRP calculated the
amount of secondary PM10 that could form from the reaction of ammonia slip with
NO2 and SO3 in the turbine exhaust. The calculations in Table 3-- show that
Equation (1) would produce up to 3.9 ton/yr of ammonium sulfate secondary PM10
from reaction of ammonia only with combustion byproducts. Additional ammonium sulfate would form from
reaction of SO3 in the atmosphere with any emitted ammonia. Table 4 shows that Equation (3) would
produce up to 105.8 ton/yr of ammonium nitrate secondary PM10 from
reaction of ammonia only with combustion byproducts. Some this PM10 would be deposited within the HRSG and the balance
emitted. Additional ammonium nitrate
could form from the reaction of NO2 in the atmosphere with any
emitted ammonia. This additional PM10
was not included in the Project’s emissions estimates. The PDOC’s conclusion that the nor the Project’s will
not trigger an emissions offset requirement is thus
false.
The CEC has made the same point in its PDOC
comments. The CEC staff concludes that “the
ammonia slip from the proposed MEC is 118.6 tons per year….The direct PM10 from the proposed MEC is 98.6 tons
per year which is close to the
District’s mitigation threshold [of] 100 tons per year. Considering the formation of secondary PM10,
ammonia nitrate, from the proposed project, the combined PM10 emissions will be
more than the District’s threshold and
qualify MEC to mitigate PM10.” [45]
ii. PM10
Formation Causes SCR Maintenance Problems
Some of these salts
estimated in Tables 31
and 42
deposits in the low-pressure tube sections of the HRSG. According to GE, "[a]ctual operating
experience indicates that ammonium-sulfur salt formation and boiler damage
occur without exception, when ANY sulfur bearing fuel is fired in the gas
turbine and SCR is used for NOx control.
This is not usually accounted for in BACT determinations, but adds
significant cost, and should be considered." These salts build up,
decreasing heat transfer and increasing operating costs. They also corrode the boiler tubes,
requiring periodic cleaning and periodic replacement of the low-pressure tube
sections of the HSRG. (Id., pp. 1,
3.) This affects the reliability and
maintenance costs of SCR and thus must be considered in the BACT analysis. The salt deposition problem will be
aggravated by the use of dry low NOx combustors, which increase the amount of
NO2 in turbine exhaust gases, and hence the amount of ammonium
nitrate that may form in the HRSG.
(ASME 1/1/99.[46])

iii. PM10 Formation Causes
Visibility Reduction
The fact that these reactions actually occur and cause visibility impacts is well documented in the technical literature. A noted atmospheric textbook, for example, contains this vivid description of the problem (Pitts and Pitts, 1999, [47] p. 284):
"The formation of ammonium nitrate has some interesting implications for visibility reduction. In the Los Angeles air basin, for example, the major NOx sources are at the western, upwind end of the air basin. Approximately 40 miles east in the vicinity of the city of Chino, there is a large agricultural areas that has significant emissions of ammonia...under typical meteorological conditions, air is carried inland during the day, with NOx being oxidized to HNO3 as the air mass
moves downwind. When it reaches the agricultural area, the HNO3 reacts with gaseous NH3 to form ammonium nitrate, the particles formed by such gas-to-particle conversion processes are in the size range where they scatter light efficiently, giving the appearance of a very hazy or smoggy atmosphere even though other manifestations of smog such as ozone levels may not be highly elevated."
iv. PM10
Formation Endangers Endemic Biota
The
majority of the ammonia slip reacts with NOx to form ammonium nitrate, which is
PM10. (Table 4.) This PM10 can be deposited on surrounding
hills, including Coyote Ridge to the east, Santa Teresa Hills to the west, and
Tulare Hill, located immediately north by northwestadjacent to of the site. These hills contain soils derived from
serpentine rock that support serpentine grasslands, considered a sensitive
habitat by the California Department of Fish and Game. These soils also support a high number of
rare and/or endemic plant species as well as endemic invertebrates such as the
federally threatened bay checkerspot butterfly. (PSA, pp. 361-362.)
Serpentine
soils are low in nitrogen, which restricts the growth of invasive non-native
plant species. The soils are currently
over-saturated with nitrogen and exceed levels known to alter serpentine
ecosystems. (PSA, p. 378.) Although the Applicant modelled the impact of
the Project on these soils and concluded that the increase in nitrogen would be
small, 1.56% of existing background (PSA, p. 378), the Applicant's analysis
apparently failed to include secondary PM10, most of which is ammonium
nitrate. This additional PM10 would
nearly double the Project's reported contribution to soil nitrogen. The impact of this additional ammonium
nitrate has not been evaluated and must be to fully evaluate the environmental
impacts of SCR.
E. ALTERNATIVES TO SCONOx TO ACHIEVE A NOx BACT of 1.3 ppm
There are
alternatives to SCONOx that achieve 1.3 ppm NOx averaged over 1 hour with very
low ammonia slip. A standard SCR can be
designed to achieve a NOx level of 1 ppm, comparable to SCONOx, by simply
adding more catalyst and increasing the catalyst change out efficiency. The ammonia slip can be reduced or
eliminated by using this standard system by either designing the SCR to achieve
a lower ammonia slip and by following the SCR with a CatCO oxidation catalyst
to remove ammonia. These two options
are discussed below.
1. Standard SCR Designed For A Lower NH3
Slip
Lower slip levels can be readily and inexpensively achieved using a standard SCR system designed to meet a lower slip. The CARB Guidance Document[48] recommends a slip of less than 5 ppm and acknowledges that slips as low as 2 ppm can be achieved using standard technology. (CARB 9/99, [49] pp. 25-26.)
Very low slips have been achieved in practice at large natural gas-fired turbines that comply with vendor-recommended maintenance (e.g., annual catalyst washing.) The Hitachi NOx guarantee letter in Appendix D of the CARB Guidance Document, for example, identifies a 1400-MW plant consisting of four GE Frame 9 gas turbines that is currently operating at a NOx level of 3 ppmvd with a 3-ppmvd ammonia slip in Japan.
Both Massachusetts
and Rhode Island have established 2-ppm ammonia slip BACT limits for new power
plants. Rhode Island requires all power
plant permit applicants to justify why they cannot achieve a 2ppm ammonia slip
for SCR as part of their BACT analysis.
The Massachusetts Department of the Environmental Protection
("MDEP") has established a “Zero Ammonia Technology” BACT standard
for gas turbines larger than 50 MW.
(Struhs 1/29/99.[50])
Three large projects in the Massachusetts market, the 350-MW Cabot Power Island End Project, the 420-MW American National Power Blackstone Project, and 1,550-MW Sithe Mystic Development have been issued PSD permits specifying a NOx limit of 2 ppm achieved with a 2 ppm ammonia slip, demonstrated using an ammonia CEMs and both averaged over 1 hour. Their permits further require that they retrofit with zero ammonia technology at the end of five years.
All of the major
SCR vendors will guarantee ammonia slips substantially below 10 ppm. Attachment D to the CARB Guidance Document
includes performance guarantees from four of the major SCR vendors for a 5-ppmv
slip, the only level requested. A slip
level of 5 ppm is currently proposed for the Moss Landing Project.[51] The CEC
staff has preliminarily called for a 5-ppmv ammonia slip limit for MEC. [52]In
addition, all of the major vendors are currently offering performance
guarantees of 2 ppmvd to compete in the New England market.[53]
Finally, the CARB
Guidance Document recommends “that districts consider establishing ammonia slip
levels below 5 ppmvd at 15 percent oxygen in light of the fact that control
equipment vendors have openly guaranteed single-digit levels for ammonia
slip.” (CARB 9/99, p. 26.)
2. SCR Plus Downstream Oxidizing Catalyst
A standard SCR
system can be designed to include an oxidizing layer downstream of the SCR
catalyst. The oxidizing layer would
oxidize ammonia to nitrogen gas and water.
However, depending on the temperature in the oxidation zone, some NOx
could be created, requiring an increase in the volume of the SCR catalyst to achieve
equivalent NOx without the oxidation zone. Near-zero slip levels can be readily and inexpensively achieved
using this system. This approach is
routinely used to control unburned hydrocarbons and CO from diesel engines
(Durilla 5/99[54]) and
represents standard practice in Europe for engines. This technique was also employed in Grace Dual Function
catalysts, which are currently operating on many large natural gas fired
turbines.
The Applicants'
consultant has argued that "if the oxidation catalyst were located
downstream of the SCR catalyst, you run the risk of converting the ammonia slip
back to NOx. Even typical ammonia slip
levels of 1-2 ppm from a new SCR catalyst would increase NOx levels by 50%-100%
from new generation facilities that are equipped with dry low-NOx combustors
and high efficiency (90%) SCR systems."
(Rubenstein 6/8/99.[55]) However, according to Englehard research
engineers, this is incorrect. The
maximum conversion of ammonia to NOx that could occur in the region where the
downstream Camet catalyst would be located is 20% and actual values would be
substantially smaller. Assuming a
typical slip level of 2 ppm, the maximum increase in NOx would be 0.4 ppm,
which would increase NOx from 2.5 ppm at the outlet of the SCR to 2.9 ppm or by
about 20%. This increase in NOx could
be readily addressed by simply increasing the volume of SCR catalyst.
IV.
BACT
HAS NOT BEEN REQUIRED FOR CO – LOWER CO LEVELS
HAVE BEEN ACHIEVED
The PDOC concludes that
BACT for carbon monoxide (“CO”) is an emission limit of 10 ppm at 15% O2
averaged over 3 hours achieved without an oxidation catalyst. However, the District failed to perform a
top-down BACT analysis, as discussed above.
Further, it made several errors in applying its own BACT
guidelines. The District argues that
its BACT determination for gas turbines larger than 23 MMBtu/hr (Guideline
89.2.1) does not apply to Metcalf because Crockett has not achieved its permit
limit and lower limits recently permitted for other similar projects have not
yet been achieved in practice. The
District's arguments are technically incorrect, violate federal BACT guidance,
and ignore significant data to the contrary.
As a result, the District has failed to establish the proper BACT level
for CO. The proposed 10-ppm limit is
much higher than CO concentrations that are routinely achieved on similar
plants and, thus, is not BACT.
A.
Lower CO Levels Have Been
Achieved
The District's BACT Guideline
89.2.1 establishes a BACT limit of < 6 ppm or 90% CO reduction as technologically
feasible and cost effective and 10 ppm as achieved in practice, both achieved
with an oxidation catalyst.[56] The District has declined to adopt the <6
ppm limit or an oxidation catalyst, contrary to its own guidance.[57]
First, the District argues that Los
Medanos, which was permitted by the District at 6 ppm CO, is not relevant to
Metcalf because it has not yet been demonstrated consistently under actual
operating conditions. (PDOC, p. 10.) This is inconsistent with U.S. EPA guidance,
which only requires that a limit be specified in a permit to be selected as the
top technology in the top-down BACT process.
Other recently permitted projects in California, outside of the
District, have been permitted at 6 ppm (e.g., La Paloma, Sunrise, Elk
Hills). Further, other states have
required much lower CO limits (2 ppmv averaged over 1 hour) in recently issued
PSD permits. These lower limits, which
have been established in valid PSD permits, should have been included in a
proper top-down BACT analysis. There is
no obligation under federal law to constrain a BACT determination to the
boundaries of the permitting agency, as done here. BACT knows no boundaries, and, in fact, EPA acknowledges foreign
experience as relevant.
Second, the District argues that
a limit lower than 10 ppm has not been achieved for the type of project (power
augmentation, duct burners, merchant operation) proposed by Metcalf,
specifically citing the compliance problems at Crockett. (PDOC, p. 10.) However, the Crockett experience is irrelevant as discussed
below. Further, CO levels much lower
than 10-ppm have been achieved in practice elsewhere, as also demonstrated
below. Finally, the specifics (e.g., duct
burners, steam injection, merchant mode) of the Metcalf Project do not affect
the performance of catalytic systems.
In these processes, simply simply adding more catalyst to reduce space
velocities can increase removal efficiencies.
Therefore, even if duct burners and steam injection for power
augmentation appreciably increased CO levels, which they apparently do not, it
would not affect the performance of a properly designed catalytic process. The increase in CO, if any, could be removed
by simply designing the system to include the proper amount of catalyst require
to achieve the target emission limit.
Under the PSD program, BACT is
“an emissions limitation . . . based on the maximum degree of reduction for
each pollutant subject to regulation under [the] Act which would be emitted
from any proposed major stationary source . . . .” (40 CFR § 52.21(b)(12),
emphasis added.) The information
reviewed below indicates that 0.5-ppm CO averaged over 3 hours is achieved in
practice. Therefore, the District has
failed to specify BACT for this Project.
A. Crockett Experience Is Irrelevant
The Crockett
facility has been unable to meet its 5.9-ppm CO limit during minimum load under
ambient conditions of low temperature and high relative humidity and during
peak load under ambient conditions of high temperature and moderate to high
relative humidity. The District uses
the experience at Crockett to argue that its BACT guideline is not
relevant. (PDOC, p. 10.)
However, the gas
turbine at Crockett is recognized by CARB “as being somewhat unique, since it
is an early version of the General Electric Frame 7FA” which has since been
discontinued. The rotor was recently
replaced due to vibration and blade erosion problems. The combustor has also been replaced, with subsequent CO emissions below 10 ppmv.[58] In CARB’s opinion, the compliance problems
at Crockett “appear to be related specifically to this gas turbine or overall
system and not necessarily to the oxidation catalyst.” CARB specifically cautioned that this
experience is unique and should not be applied to other projects, as advocated here
by the District. (Menebroker 9/1/99.[59])
Further, Engelhard, the vendor of the oxidation catalyst, has conducted extensive investigations into the possible causes of the CO compliance problems at Crockett. Contrary to the District's claims, it has concluded that the compliance problems are unlikely to be related to either the weather or the catalyst itself (plug samples consistently showed the catalyst was good), a similar catalyst at the same site was operating properly, and numerous similar facilities are in successful operation worldwide. (Mack 7/19/99.[60]) There are other, more plausible explanations, not related to the efficacy of the catalyst itself. These include sugar deposits (from the adjacent C&H sugar plant), catalyst plugging from internal insulation sloughing,[61] and turbine problems. Therefore, the District has inappropriately used the Crockett experience to establish a CO BACT limit for Metcalf that is higher than CO levels achieved in practice at other power plants.
B. Source Tests Support A CO Limit <1 ppm
The information compiled in the
CARB Guidance Document supports a much lower BACT level than 6 ppm averaged
over 3 hours. For combined-cycle
plants, the most stringent emission limit for CO required in a construction
permit is 1.8 ppmvd at 15%O2 averaged over 1 hour at the Newark Bay
Cogeneration facility. (CARB 9/99,
Table C-6.) Compliance with this limit
was demonstrated in a source test.
(Id., Appx. C, p. 26.) Moreover,
the actual CO concentrations measured in all nine of the CO source tests
summarized in the Guidance Document are at or below 2.0 ppmvd at 15% O2. (Id., Table C-8.) These data support a BACT level for CO for combined-cycle plants
of 2.0 ppmvd @ 15% O2 averaged over 1 hour.
In addition, source test data for
four additional combined cycle and cogeneration gas turbines equipped with
oxidation catalysts (Table 5) support a CO BACT level of less than 1 ppm. Table 5 includes six sets of source tests
that were performed at loads ranging from 50% to 100%. These partial load data are representative
of merchant operation and indicate that CO (and VOC – see Table 5) levels
during partial load operation are comparable to those during full load
operation. Most of the CO measurements,
irrespective of load, are much less than 1 ppm. These data confirm that BACT for CO for large combined cycle gas
turbines in merchant operation is no more than 2.0 ppmvd @ 15% O2
averaged over 1 hour and support a BACT CO level of less than 1 ppm.
C. SCONOx Achieves 0.7 ppm CO
SCONOx simultaneously removes NOx,
CO, and VOCs. The nine months of recent
CEMs data discussed above in Comment III.B.1 indicate that the Federal Facility
routinely achieves a CO limit of 1.0 ppm averaged over 1 hour, and 0.7 ppm
averaged over 3 hours. Similar performance
has been demonstrated at the Genetics facility. The 1-hour average CO data are summarized in Figure 2, and the
3-hour average data in Figure 3. These
figures exclude excursions due to startup, shutdown, and non-SCONOx operating
problems (e.g., CEM failures, plant trips, operator error, condensation in gas
generator).
The Applicants have argued that
duct firing would somehow limit SCONOx's ability to control CO. (Metcalf Response to CVRPCVRP Data Request 8e.) This is erroneous. SCONOx has been operating for nearly a year at the Genetics
Institute in Andover, Massachusetts, which employs a duct-fired heat recovery
steam generator (“HRSG”). Because both
the turbine and the duct burners burn natural gas, the emission characteristics
are very similar. In any event, the
duct burner emissions comprise only a small fraction of the total exhaust
gases, 7% in the case of Metcalf that would have to be treated by a SCONOx
system. This small increase would not
alter the system’s fundamental design.
Finally, the vendors of SCONOx have confirmed these facts and have
further noted, based on experimental tests, that duct firing actually improves
the performance of SCONOx, not reduces it as alleged by the Applicants.
The Applicants have also argued
that "the Applicant is not aware of the performance of the SCONOx system
in combination with the inherently low levels associated with the advanced
combustors proposed for use at MEC."
(Metcalf Response to CVRPCVRP
Data Request 8a, p. 9.) The type of
combustor is irrelevant to the performance of SCONOx. The only important variable is inlet CO concentration. ABB guarantees a CO reduction of 90%,
irrespective of the inlet concentration.
Therefore, for Metcalf, SCONOx could be designed to achieve 1.0 ppm CO,
not 2 ppm as alleged by the Applicants.
(Id., p. 10.)
D. An Oxidation Catalyst Achieves 0.5 ppm
Continuous Emission Monitoring (“CEM”) data from the Southwest Air Pollution Control Agency (“SAPCA”) in Washington for the River Road Generating Project indicates that this facility, which is a large Frame 7, meets a CO limit of 0.5 ppm averaged over 3 hours. The facility is a 248-MW natural gas fired, combined-cycle plant consisting of a GE 7231 FA gas turbine equipped with GE dry low-NOx combustors (DLN III), an unfired HRSG, and a steam turbine. Control equipment includes an SCR system permitted at 4 ppmvd NOx at 15% O2 averaged over 24 hours, and a CO oxidation catalyst guaranteed by the vendor at 3 ppmvd at 15% O2 and permitted at 6.0 ppmvd at 15% O2 averaged over 3 hour. The unit operates at loads from 75% to 100%, and experiences frequent shutdowns and startups.
The 1-hour averaged CEM data for the last four quarters of operation are included in (05-31-00 CVRP comments on MEC PDOC: Exhibit 16). These data indicate that the River Road Generating Station routinely achieves a CO limit of 1.2 ppm averaged over 1 hour and 0.5 ppm averaged over 3 hours. The 1-hour average data are shown in Figure 4 and the 3-hour average data in Figure 5. All exceedances of these limits were due to startups, shutdowns, operator error, or equipment malfunctions reported to the SAPCA, with the exception of a single event on 12/20/98 from 12:00 PM to 1:00 PM. Although not of a Westinghouse 501 F equipped with dry low NOx combustors can meet 10 ppm CO for all operating conditions proposed by Metcalf. In fact, Metcalf has agreed to 10 ppm for all operating conditions. (PDOC, p. 11.) Oxidation catalysts can be readily designed to remove 90% or more of the CO. (Mack 7/19/99.[62]) Therefore, it is clearly feasible to meet a CO limit of 1.2 ppm averaged over 1 hour or 0.5 ppm averaged over 3 hours, consistent with the River Road data.
V.
catalytic
controls do not significantly increase pm10
The Applicants argue that both SCONOx and oxidation catalysts
significantly increase PM10 emissions, thus offsetting the benefits of CO
reduction because CO is attainment and is therefore safe, and PM10
is nonattainment in the District. This
is untrue on both countswith regard to both SCONOx and oxidation catalysts,
as discussed below.
A.
An
Oxidation Catalyst Does Not Increase PM10
The Applicant, in response to
District inquiry, argues that an oxidation catalyst should not be used because
80% of the SO2 is oxidized to sulfate, increasing PM10 emissions. (Rubenstein 6/14/00.[63]) The Applicant supports its argument by
attaching a memorandum from its consultant to CARB. (Rubenstein 6/8/99.[64])
The Applicant’s consultant has
made this argument in other
forumselsewhere, where it
has been repeatedly rejected. The
proffered letter was originally submitted to CARB during development of the
Power Plant Guidance Document in an attempt to persuade CARB not to endorse
oxidation catalysts.[65] CARB rejected these arguments, concluding
that "[f]rom the perspective of staff, there is not enough evidence
indicating any significant increase PM10 emission caused by oxidation
catalysts." (CARB 9/99, Appx. C,
p. 29.) Engelhard, the largest
manufacturer of oxidation catalysts, also previously rebutted these
claims. (Mack 6/1/99.[66])
In response to comments filed by
others, the Applicant’s consultant subsequently claimed that a “confidential”
source test supported his position that oxidation catalysts generate PM10.[67] These subsequent claims were likewise
rebutted by both CARB (Menebroker 1/7/00[68])
and Engelhard. (Mack 12/22/99.[69]) CARB, for example, concludeds: “[w]e do not believe that there is
adequate evidence proving that oxidation catalysts contribute to PM10 emissions
from gas turbines,” confirming their original conclusion in the Guidance
Document. Engelhard concurred: “the
challenges against CO oxidation catalysts raised in the Sierra Environmental
letter regarding PM10 are inaccurate and unsubstantiated,” pointing
out that if the “confidential” source test actually does support PM10
generation, the PM10 is likely from the SCR system, which is a well
known source of PM10 caused by the oxidation of sulfur across the
SCR catalyst.
In fact, the claims made in the June 8, 1999 Rubenstein letter submitted to the District are incorrect.
First, they are based on a 1993 engineering estimate, which is not provided. The catalyst industry has changed dramatically since 1993 and even if the analysis were then accurate (for which there is not evidence), it would not be accurate today.
Second, the letter claims conversion of SO2 to particulate sulfate (SO4), based on an attached Grace curve. However, the curve is for conversion of SO2 to SO3 as a function of gas temperature, not of SO2 to SO4. To form particulate matter, any SO3 that is formed across the catalyst would have to combine with moisture in the stack gas to form H2SO4 and then with ammonia to form ammonium sulfite and ammonium bisulfite to form particulate. Third, the proffered curve is for a space velocity of 108,000 per hour. Catalyst removal efficiency is directly proportional to space velocity, which is the ratio of exhaust gas flow rate through the catalyst to catalyst volume, while SO2 oxidation is inversely proportional to space velocity. Oxidation catalysts today are designed to operate at a space velocity of 200,000 per hour, not 108,000 per hour. The conversion of SO2 to SO3 increases as the space velocity decreases. Therefore, the proffered curve, even if it were relevant, overstates the conversion, if any.
Third, the letter claims 80%
conversion based on the Grace curve. An
inspection of this curve indicates that 80% conversion corresponds to a
temperature of about 1000 F. However,
oxidation catalysts are typically installed in the HRSG where the temperature
is 600 to 650 F, not at 1000 F.
Similar curves, supplied by the
largest manufacturer of oxidation catalysts in the world, indicate that the
conversion of SO2 to SO3 at a space velocity of 200,000
per hour at 650 F, the highest temperature likely to be experienced, would be
about 5%, not 80% as claimed by the Applicant.
To contribute to particulate matter in the stack, this SO3
would have to react with ammonia and condense.
Condensation would only occur if the dew point of the ammonia reaction
products is above the exhaust temperature at the stack. The dew point of sulfate reaction products
is about 120 F, well below the stack temperature of combined cycle plants,
including the Metcalf facility.
Therefore, the oxidation of SO2 to SO3 will not
form particulate matter. Further, even
if condensation occurred, the presence of ammonia from the SCR system is the
culprit, not oxidation of SO2 to SO3 across the oxidation
catalyst. Ammonia can be substantially
reduced by using lower the slip or eliminated using a downstream ammonia
oxidation step, as discussed in Comment --.
Finally, we note that the
Applicant has argued that oxidation catalysts increase PM10 while ignoring the
identical problem for the SCR. However,
in the case of NOx , which is also an attainment pollutant, the
Applicant has advocated an SCR system without any concern for the very same PM10
issue. SCR systems generate substantial amounts of PM10 from
oxidation of SO2 to SO3 and from secondary PM10
formation from the ammonia, as discussed above in Comment --.
In sum, there is no credible
evidence that oxidation catalysts generate PM10. This is a non-issue, which should not be
considered in the District’s BACT analyses.
B.
IF AN OXIDATION CATALYST WOULD INCREASE PM10, THEN
MEC REQUIRES PM10 OFFSETS
The PDOC states that a CO catalyst will not be part of the initial control technologies employed. But if the system does not achieve the CO emissions requirement (which is 10 ppm in proposed Condition 20(d) of the PDOC, but which we argue in our comments should be much lower), then MEC would be required to install an oxidation catalyst according to permit condition 23. However, according to Calpine consultants, the use of an oxidation catalyst may well increase the PM10 emissions significantly, and to the point where the emissions would exceed the 100-tpy threshold. Because offsets would then be required, and because offsets are difficult to obtain, this contingency must be addressed up-front, at the permitting stage. The District must either explicitly reject the argument that CO catalysts increase PM10 emissions, or if it does not it must require that PM10 offsets be acquired because of the PM10 emissions implications of licensing condition 23.
References provided are from submissions made by Calpine to the Connecticut DEP for the Towantic Energy Project.) The PDOC states that a CO catalyst will not be part of the initial control technologies employed. The statement is that the MEC will attempt to achieve a stack gas concentration of 10 ppm of CO without the use of a catalyst. If the system does not achieve the 10ppm requirement, the MEC would install an oxidation catalyst according to permit condition 23. However, the use of an oxidation catalyst may well increase the PM emissions significantly, and to the point where the emissions would exceed the 100-tpy threshold. Because offsets may then be required, and because offsets are difficult to obtain, this contingency must be addressed up-front, at the permitting stage. Therefore, the projected increase of PM emissions associated with the potential use of an oxidation catalyst needs to accounted in the estimate of total PM emissions. As is discussed elsewhere, the emissions of PM have been already underestimated by a factor of two because of the omission of the condensable fraction of PM in the emission calculations. The use of the catalyst in the secondary formation of particulates will further increase the total emissions of particulates.
Attached are tabulated values of the PM emissions estimated by R.W.Beck, the air quality consultant to Calpine’s Towantic Energy Project in Oxford, CT. Table 6-6, dated 12/22/1998 shows that the PM emissions were originally in the range of 2.6 g/s for natural gas combustion for a GE 7241 (FA) combustion turbine with HSRG. Table 6-6 (Revised), dated 10/25/1999 shows that when an oxidation catalyst is employed to reduce CO emissions, the PM emissions associated with natural gas firing increase to about 4.4 g/s. This is a 50 % increase. The excerpt from the enclosed transmittal letter to Mr. Sinclair and footnote 6 to the revised table document the association of the increases to the use of the CO catalyst.
When the increase of emissions of PM10 is calculated, it will be necessary to recalculate the Significant Impact Area surrounding the site. Emissions from other sources within this area will have to account for directly in the ambient air quality compliance computations. It is certain that an increase of PM10 emissions from the proper estimate of the formation of condensable particulates will push the present peak prediction of 9.3ug/m3 well over the monitoring significance level of 10ug/m3 for PM10.
C.
SCONOx
Does Not Increase PM10
The Applicant has also claimed in
the Energy Commission proceedings that Nueva Azalea, which is using SCONOx to
control CO and NOx, would emit "32.8 lbs/hr during base load operation,
more than three times the comparable figure for MEC...We believe that it would
be a poor trade-off indeed to reduce CO levels (which are already safe) and
nearly triple PM10 emission rates for the sake of "matching the
performance" of another project."
(Metcalf Response to CVRPCVRP
Data Request 8e.) This is unsupported
and incorrect.
The PM10 emission rate that is
being permitted in the Nueva Azalea case is 13.6 lb/hr per turbine, not 32.8
lb/hr as claimed by the Applicants.
(Nueva Azalea AFC,[70]
Table 5.2-22.) This rate is only 13%
higher than the PM10 emissions being permitted by the Applicant. However, this comparison is irrelevant for
three reasons.
First, Nueva Azalea is using AB24
gas turbines, while the Applicants are using Westinghouse 501F turbines. These two turbines have different exhaust
PM10 emission rates, totally independent of installed controls. Thus, they cannot be directly compared to
evaluate the contribution of SCONOx as advocated by the Applicants.
Second,
as discussed in Comment --, the Applicants have understated their PM10
emissions. Actual vendor-guaranteed
emissions for the Applicants' Westinghouse 501F turbines are 22 lb/hr, not the
12 lb/hr that is being permitted.
Third, it is chemically and
physically impossible for SCONOx to generate substantial amounts of PM10. First, SCONOx in some applications uses an upstream SCOSOx
catalyst, which removes over 95% of the SO2 from the gas stream. The
SO2 is subsequently stripped from the SCOSOx catalyst during
catalyst regeneration and is either removed by a solid scrubber or routed
around the SCONOx catalyst, thus eliminating any possibility of oxidizing over
95% of the SO2 to SO3.
Second, SCONOx uses no ammonia, and, as discussed above, ammonia is
necessary to convert any SO3 to particulate. Thus, even if small amounts of SO3
formed, it would not react with ammonia to form particulate.
Finally, actual source tests on
the Federal facility demonstrate that SCONOx (in
this case without use of SCOSOx) does not increase PM10. (Delta 2/9/98.[71]) Two identical LM2500 turbines, one with
(Federal) and one without SCONOx (Growers), were tested. The PM10 emissions from the SCONOx facility
(0.0003 g/dscf or 0.28 lb/hr) are half of those from the non-SCONOx facility
(0.0006 gr/dscf or 0.60 lb/hr).
Further, the PM10 concentration achieved in the SCONOx test (0.000038
lb/MMBtu) is substantially smaller than the PM10 limit proposed by Metcalf
(0.00565 lb/MMBtu). Therefore, the
claim that SCONOx increases PM10 is wrong.
VI.
OTHER AIR QUALITY The Benefits Of CO control
The Applicant's consultant has
argued that CO should not be controlled because stack concentrations are lower than
ambient air quality standards and hence safe.
This argument turns the PSD program on its head by advocating for no CO
control when an area is in attainment.
Further, the NSR Manual explicitly requires that the environmental
tradeoffs of control technologies be evaluated. In this case, the failure to control CO has the very real
potential of increasing ozone, which would aggravate the Bay Area's existing
ozone compliance problems. Two
significant benefits of controlling CO using either an oxidation catalyst or
SCONOx are discussed below.
A.
CO
Control Simultaneously Removes VOCs, Which Are Ozone Precursors
The SCONOx and CO catalysts both
consist of a ceramic substrate impregnated with platinum and is essentially
oxidation catalysts. The SCONOx system
achieves higher CO removal efficiencies because it operates at much lower space
velocities, typically 22,000 per hour compared to 200,000 per hour for an
oxidation catalyst. Therefore, some
collateral VOC (and toxics) reduction occurs across both catalysts, depending upon their operating
temperature, which areis determined by theirits placement in the HSRG.
The collateral VOC (and toxics) reduction for a conventional CO catalyst could be as high as 50% of the quoted CO reduction, depending upon catalyst operating temperature and the composition of the exhaust gas stream. (Heck and Farrauto 1995, Chapter 11.) Aldehydes, alkanes, alkenes higher than butane, and aromatics, such as benzene, are readily oxidized across Engelhard's CO catalyst. Most of the specific organic compounds found in turbine exhaust fall into these classes.[72] Reported removal efficiencies range from 71% for toluene to 86% for acetylene. (Heck and Farrauto 1995, Table 11.1.) Similarly, source tests at the Federal Facility, a low-temperature retrofit application, indicate that SCONOx reduces formaldehyde by 97% and acetaldehyde by 94%. (Delta 4/2/97;[73] Delta 4/00.)
Generally, the higher the
temperature, the higher the collateral VOC reduction. For example, the vendor guaranteed a 30% non-methane, non-ethane
hydrocarbon[74] reduction
and 80% CO reduction for the Tenaska 248 MW GE Frame 7FA combined cycle plant
with the catalyst located in a high temperature zone of 1000 to 1100ºF. The guarantee letter is attached in
(05-31-00 CVRP comments on MEC PDOC: Exhibit 39).[75] Although we do not know the precise location
and hence gas temperature where catalyst would be located in the Metcalf HRSG,
it likely would be located in the high temperature zone of the HRSG where the
temperature is 600-650 F. Therefore,
high collateral VOC reductions of at least 30% and perhaps as high as 50% could
be readily achieved for Metcalf using a conventional oxidation catalyst and
substantially higher for SCONOx.
B.
CO
Itself Is An Ozone Precursor
CARB and EPA Region
9 have historically declined to establish a lower CO BACT level than 6 ppm
averaged over 3 hours because CO is attainment in most of California, most of
the ambient CO is caused by motor vehicles, and the current BACT level is less
than the ambient air quality standard on CO (see CARB 9/99, p. 29). However, these are not valid reasons under
the federal definition of BACT to decline to establish a valid BACT limit.
Carbon monoxide is oxidized in
the atmosphere to ozone.[76] Generally, the ozone formation potential of
CO compared to most VOCs is quite low, but varies with atmospheric composition.[77] The ozone formation potential of CO is only
5% to 20% of that of poorly reactive alkanes (ethane, propane) and alcohols,
and substantially less for more reactive compounds such as aromatics and highly
reactive alkanes. Because CO has a low
ozone formation potential on a per weight basis, EPA and other regulatory
agencies do not consider CO to be an ozone precursor and have exempted it from
ozone precursor status. However,
ambient CO concentrations in much of California are typically several parts per
million – considerably higher than precursor VOCs, which are collectively
present at several hundred parts per billion of carbon. Consequently, despite its low specific
reactivity, CO may still contribute significantly to the formation of
ozone. This is a particularly important
issue in the Bay Area, which was recently re-designated by EPA as nonattainment
for ozone. Further, the NSR Manual
requires that ozone precursors be evaluated in the environmental analysis
required under the top-down BACT analysis.
Two studies have modeled the
contribution of VOCs and CO to ozone formation at various VOC/ NOx
ratios.[78] The results are summarized in Table 5.
In the Los Angeles study, the
initial composition of the atmosphere was selected to represent summertime
conditions in Los Angeles -- 1,100 ppb of nonmethane hydrocarbons (“NMHC”) and
1,500 ppm of CO expressed as carbon.
The results indicate that CO contributes 5% to ozone formed under
conditions representative of the Los Angeles Basin. (Exhibit 40.)[79]
In a similar study in Atlanta,
initial conditions were selected to represent typical center city values -- 816
ppb of VOCs and 1,200 ppb of CO expressed as carbon. CO contributed 17.5% to the 209 ppb ozone peak. (Exhibit 41.)[80] The considerably larger contribution of CO
to ozone in Atlanta compared to Los Angeles is likely due to several reasons: First, the initial CO/NMHC ratio was higher
in Atlanta (1.47 vs. 1.36). Therefore,
CO accounted for a somewhat larger fraction of total carbon in Atlanta than in
Los Angeles. Second, the Atlanta model
included a gradual increase in mixing height during the day, as well as the
presence of 500 ppb of CO aloft, compared to 30 ppb of VOC aloft. Thus, mixing caused greater dilution of VOC
than CO. Third, the VOC composition in
Atlanta contained lower fractions of highly reactive species, particularly of
alkenes. This would tend to reduce
overall ozone production, and also increase the relative importance of less
reactive species, such as CO, methane, and alkenes.
Thus, while CO is a relatively
weak ozone precursor compared to many organic compounds such as alkenes, the
concentration of CO is substantially higher than other precursor
compounds. Therefore, as demonstrated
by the two studies summarized in Table 5, CO can contribute substantially to
atmospheric ozone.
The relative ozone formation
potential of CO from Metcalf can be estimated and compared to that of VOC
emissions using the relative reactivity scale developed and routinely updated
by Carter. (Exhibit 42.)[81] The “incremental reactivity” of a VOC (grams
of ozone per gram of VOC) has become an established method of quantifying and
comparing ozone formation potential under specific atmospheric conditions. Thus, the VOC equivalents of CO emissions
can be conservatively estimated from the incremental reactivity of CO relative
to propane, the least reactive compound considered a VOC by EPA.
Most recent reactivity data
(Exhibit 42) indicate that the mass-based reactivity of propane exceeds that of
CO by a factor of 9 to 10. The ratio of
reactivity of two different ozone precursors varies with atmospheric
conditions, particularly the VOC/ NOx ratio.[82] However, the relative reactivity of poorly
reactive compounds, such as CO and lighter alkanes, such as propane, are not
very sensitive to atmospheric composition.[83] Thus, using a VOC-equivalency factor of 0.1
for CO represents the most conservative choice for assessing the relative ozone
formation potential of CO emissions because it is based on propane. Consequently, the ozone-forming potential of
permitted CO emissions from Metcalf of 735.1 ton/yr (PDOC, Condition 25, p. 29)
corresponds to about 74 ton/yr of VOC emissions or nearly 1.5 times more VOCs
than the Project would emit directly (49 ton/yr).
The actual contribution of CO to
ozone formation, compared to more reactive VOCs, could be larger because the
reactivity scales do not consider the fact that poorly reactive compounds such
as CO have a longer effective residence time in the atmosphere, compared to
more reactive species which are more rapidly converted, which may increase
their total yield compared to more reactive compounds. Thus, during inversions and other conditions
when atmospheric dispersion is poor, CO and alkanes will contribute, on a per
mass basis, more to ozone formation than projected from their incremental
reactivity. An assessment of the impact
of such conditions on the relative ozone formation potential of CO can only be
evaluated by modeling on a case-by-case basis.
However, the above discussion suggests that CO emissions from Metcalf
would contribute emissions of VOC equivalents of at least 74 ton/yr.
The formation of ozone precursors
must be considered in the environmental analysis required as part of the
top-down process. In fact, the NSR
Manual specifically contemplates that precursor compounds be considered in the
BACT analysis, as is demonstrated by the following statement:
“For
example, the use of certain volatile organic compound (VOC) control
technologies can increase nitrogen oxides (NOx) emissions. In this instance, the reviewing authority
may want to give consideration to any relevant local air quality concern
relative to the secondary pollutant (in this case NOx in favor of one having less of an impact on
ambient NOx concentrations.”
(NSR Manual, p. B.49.)
In sum, Metcalf’s CO emissions
would result in the formation of about 79 ton/yr of ozone in an area that
currently violates the federal and California ozone standard. Thus, a CO BACT limit more stringent than 10
ppm is warranted.
VII.
VOC
Limit is not BACT
The District established BACT for
PVOCs as 2 ppmvd @ 15% O2
averaged over 1 hour based on CARB's Guidance Document. In making this determination, the District
erroneously eliminated from consideration or overlooked source tests that
suggest that BACT for PVOCs is lower. (PDOC, p. 11.) Substantial evidence, not considered by the District,
demonstrates that 2 ppmv is not BACT for the Project’s PVOC emissions. The evidence reviewed below collectively
indicates that BACT for PVOCs is a limit of
no more than 0.1 ppmv based on the Crockett and River Road source tests.
In spite of its CO compliance
problems, the Crockett facility achieved 0.007 ppm in one test and 0.041 in the
other (CARB 9/99, Table C-12). These
source tests show that emission levels are much lower than the BACT level of 2
ppm selected by the District as BACT for PVOC. The low VPOC
emission levels achieved at Crockett were confirmed at the River Road
Generating Project, a 248-MW GE Frame 7FA turbine that source tested at 0.0 ppm
[sic] at 15% O2. (Ex.
25.) These two units are comparable to
Metcalf with respect to size, operation mode, and fuel composition. Thus, the District should have selected a
much lower VPOC limit than 2 ppm as BACT for Metcalf.
The CARB Guidance Document also
reports much lower achieved in practice PVOC limits including
<0.8 ppmv at Bear Mountain and <0.67 to <0.71 ppmv at Brooklyn Navy
Yard. Although the turbines are smaller
than proposed by Metcalf, the size and load of the turbine do not affect the
ability of an oxidation catalyst to control either CO or VPOCs. This is demonstrated by four sets of source
tests in Exhibit 25, which were conducted at both 50% and 100% loads. These tests show comparable VPOC levels and performance of the oxidation
catalyst at both loads. The PVOC (and CO) limit that can be
achieved by an oxidation catalyst depends on the stack gas composition and the
design of the oxidation system, not on the size of the turbine or its operating
mode. The operating mode is normally
accommodated in the design of the oxidation catalyst by simply increasing the
volume of catalyst to control potential excursions during low load operation.
Similarly, the District ignored
the lower PVOC BACT limits that have been
established for Calpine’s Sutter Power
Project (1.0 ppmv) and for La Paloma
Generating Company (1.1 ppmv). As
discussed above, to qualify for inclusion in the top down process, an emission
limit need only be specified in a permit, not demonstrated in practice.
Therefore, the District should have explicitly considered these lower permit
limits in its BACT decision.
VIII. startup and shutdown
emissions are improperly permitted
The District did
not consider startup and shutdown emissions in its BACT analysis. Further, the proposed limits are
inconsistent with vendor data. These three two issues are discussed below.
A.
BACT
Has Not Been Required For Startup And Shutdown Emissions
During startups and shutdowns,
combustion temperatures and pressures change rapidly, resulting in inefficient
combustion and higher emissions of NOx, CO, and VOCs than during
steady state operation. Further, during
much of this transient period, the flue gas temperatures are lower than the
design temperature of the SCR and oxidation catalysts, reducing their removal
efficiency and further increasing emissions.
The U.S. EPA has consistently defined startup and shutdown to be part of the normal operation of a source. (Bennett 9/28/82,[84] 2/15/83.[85]) The U.S. EPA has also consistently concluded that these emissions “should be accounted for in the design and implementation or the operating procedure for the process and control equipment. Accordingly, it is reasonable to expect that careful planning will eliminate violations on emission limitations during such periods.” (Ibid.) Furthermore, the new source performance regulations under the Clean Air Act provide as follows:
At all times,
including periods of startup, shutdown, and malfunction, owners and operators
shall, to the extent practicable, maintain and operate any affected facility
including associated air pollution control equipment in a manner consistent
with good air pollution control practice for minimizing emissions. (40 CFR § 60.11(d).)
CARB has also stated that “the
BACT decision should consider control of emissions during such periods of
operation.” (CARB 9/99, p. 34.) Hence, the Project’s startup and shutdown
emissions should be considered in the BACT analysis, and all reasonable
measures should be taken to minimize these emissions. (Rasnic 1/28/93.[86])
The draft permit in the PDOC
includes separate limits for NOx and CO for hours in which startups
and shutdowns occur. (Draft Permit,
Condition 2137.)
The EPA does not generally concur with this method of limiting startup
and shutdown emissions. (Rasnic
1/28/93, p. 2.) The record contains no
evidence that the Project’s startup and shutdown emissions were evaluated in
the BACT analysis, or that efforts would be made to assure that these emissions
are appropriately controlled.
The proposed permit limits for
the Project’s startup and shutdown emissions are much higher than levels that
are routinely achieved using SCONOx.
SCONOx is capable of achieving much lower emissions during startup and
shutdown because it is fully operational at much lower temperatures, typically
300oF, than other catalytic-based systems.
The Applicant's turbine vendor
could optimize the startup/shutdown process to reduce the Project’s startup and
shutdown emissions. In addition, there
are a number of controls available to the Applicant that could be used to
satisfy BACT and reduce startup and shutdown emissions. These include the following:
· Use of an auxiliary boiler or other source of sealing steam to reduce startup time.
· Use of a stack damper to keep the HRSG hot during shutdown;
· Early injection of NH3 into the SCR;
· Use of alternatives to low-NOx combustor technology, such as XONON, which can achieve 3 ppm NOx at 15% O2 as currently proposed by Pastoria;
· Use of more efficient primary control technologies, such as SCONOx; and/or
·
Use of other methods to more quickly heat up the
catalysts in the control technologies.
(CARB 9/99, p. 35)
·
B.
Startup
And Shutdown Emissions Are Inconsistent With Vendor Data
The District
established startup and shutdown emissions in draft Permit Condition 21,
apparently calculated by multiplying the hourly startup emission rate by the
estimated startup time. (AFC, Supplement
C, Table 8.1A-2.) However, the
Applicant's data that the District relied on is inconsistent with vendor data
submitted in other cases. The following
table compares the proposed permit limits with data supplied by Westinghouse
for its 501F machine in other cases:
EMISSIONS (lbs per event per turbine)
NOx CO
VPOC
Cold Hot Cold Hot Cold Hot
TMP 556 153 1163 853 146 115
Sutter 306 170 1466 902 - -
MSEP 350 120 1825 970 200 120
MEC 240 80 2514 902 48 16
These data
indicate that the proposed permit limits for NOx and PVOCs are substantially lower than
vendor-supplied data while the CO cold start limit is substantially
higher. Therefore, the District should
request supporting vendor data and verify that the proposed permit limits are
reasonable. This is particularly critical if these emissions
are not continuously monitoredThe
District should also require that the continuous emissions monitoring cover
startup and shutdown periods, and require the Applicant to use a CEM system,
which is capable of producing accurate
measurements during the varying conditions, which will exist during startups and shutdowns.
.
.
IX.
PM10
EMISSIONS ARE IMPROPERLY ESTIMATED AND
PERMITTED
A.
The Level of
PM10 Emissions is Understated in the
PDOC
1. PM10
emissions from the gas turbine and duct burners
The PDOC is based on a PM10
emission rate of 12 lb/hr (PDOC, p. 278, Condition 20(h)),
broken down as 10 lb/hr for the gas turbine and 2 lb/hr for duct firing and
steam injection for power augmentation.
This emission rate is used to determine the need for PM10 offsets and
PSD review, both of which are triggered if emissions exceed 100 ton/yr. The total PM10 emissions, based on this
limit, are 90.6 ton/yr without emergencystandby engines or cooling towers and 98.55 ton/yr with emergency cooling towers and standby engines (PDOC,
Table 3), allowing the Project to escape offsetting its PM10 emissions and
going through PSD review for PM10.
Although the PDOC claims that this emission rate is based on a vendor
guarantee (PDOC, pp. A-4, A-5), the guarantee is not provided and the rate
itself is inconsistent with emission rates quoted by Westinghouse for other
similar sized projects.
Vendor-guaranteed emissions for
Westinghouse 501F engines for other similar projects are included here and compared to the emissions claimed by
Metcalf in the followingbelow insert table (the two lines for Metcalf are without and with
duct burners, respectively:
Fuel Vendor Adjusted
Project MMBtu/hr PM10 PM10
LHV lb/hr lb/hr
New Milford 1875 8.45 16.2
Three Mountain 1689 16.4 17.4
Midway-Sunset 1704 19.2 20.2
Elk Hills 1728 17.3 18.8
Sutter 1705 30.3 31.9
Metcalf 1794 102
Metcalf 1914 12
AVERAGE 20.1
The emissions in this table were
adjusted to a Metcalf basis (last column) to account for difference in fuel
flow. The New Milford, Connecticut PM10
data were also doubled to account for the fact that only front half
(filterable) PM10 is included in the vendor estimate. The vendor-guaranteed PM10 emissions range from 17.9 lb/hr per
turbine to 30.3 lb/hr per turbine, with the highest value being guaranteed for the nearly
identical Calpine Sutter Project with duct burners and steam injection. These emissions are substantially higher
than the 12 lb/hr claimed in the PDOC for Metcalf. Therefore, the District should support the PM10 emissions with a
valid vendor quote.
Normally, a vendor guarantee is
required to confirm that a proposed BACT limit is technically feasible. (NSR Manual, p. B.20.) There is no evidence here that a vendor
guarantee for the 12 lb/hr has been obtained.
The magnitude of the proposed limit suggests that it is based only on
front half or so-called filterable PM10.
Federal regulations require that permits be based on total PM10.
A number of power plant
applicants have recently proposed similarly low PM10 limits based only on the
filterable fraction to reduce PM10 offset liability (e.g., Sunrise, Moss
Landing), assuming that the condensable fraction of PM10 is
negligible. They are gambling that
exceedances will not be detected because only annual or less frequent source
testing is required, during which operations are typically optimized to
minimize emissions immediately prior to a source test.
We reviewed source tests of similar facilities to determine whether the proposed limit could reasonably be expected to be met (see Table 6). This review demonstrates that the PDOC’s limit is unrealistically low, unlikely to be achieved in practice, and, if based only on the filterable or front-half portion of PM10, which is about 50% of the total, is based on a faulty assumption. (Table 6.) Based on these source tests, it is likely that the proposed limit will be exceeded.
However, it is unlikely that the
exceedances will be detected, unless a continuous emission monitor is
used. It is well known that “[m]anual
stack tests are generally performed under optimum operating conditions, and as
such, do not reflect the full-time emission conditions from a source.” (40 FR 46241 10/6/75.) A widely used handbook on CEMs notes, with
respect to PM10 source tests, that: Due to the planning and
preparations necessary for these manual methods, the source is usually notified
prior to the actual testing. This lead
time allows the source to optimize both operations and control equipment
performance in order to pass the tests.”[87] Therefore, it is unlikely that violations of
the proposed 12-lb/hr limit would be detected.
The NSR Manual requires that “BACT
emission limits or conditions must be met on a continual basis at all levels of
operation...and be enforceable as a practical matter.” (NSR Manual, p. B.56.) PM10 is routinely continuously
monitored in Europe. The EPA has
required continuous PM10 monitoring on incinerators and has proposed
Performance Specification 11 for their certification.[88] Therefore, we recommend that the District
either require continuous monitoring for PM10, or require the Applicant to produce a valid
vendor guarantee that demonstrates that 12 lb/hr represents the maximum front and back half PM10 emissions from its
turbines. Source test data suggest that the proposed PM10 limit in the PDOC will be exceeded.
2. PM10 emissions from the cooling towers due to inadequate cooling tower permit conditions
The PM emissions from the 10 cooling towers contribute significantly to the ambient air concentrations of PM10 concentrations. The effluents have low exit temperatures, low exit velocities and correspondingly are low in momentum and buoyancy. When ISC 3 is used to model the impact of these emissions as point sources, as was done in the applicant’s submission, ISC3 models these as point sources and the Briggs plume rise equations are used. The plumes are predicted to have significant plume rise and the ground level concentrations are predicted accordingly. Observing that the releases often show little buoyancy, an alternative method of modeling the emissions is to assume they emanate from a series of volume sources based upon the dimensions of the individual cooling towers. This method better simulates the observation that there is little plume rise. When volume source configurations replace the point source approximations in ISC3, the predicted concentrations increase substantially, from a maximum value of about 8ug/m3 on a 24-hour basis to a value of 61 ug/ms. A value of 61 ug/m3 would constitute an exceedance of the Federal PSD increment of 30 ug/m3 for PM10. Because of the sensitivity of the predicted concentrations to the way that the cooling tower emissions are modeled, the applicant should attempt to demonstrate through alternative approaches, that the cooling towers would not cause an exceedance of a PSD increment
The
cooling tower would emit 7.95 ton/yr of PM10, calculated assuming a cooling
tower circulation rate of 133,378 gpm, a maximum total dissolved solids
("TDS") of 5,438 mg/L, and a drift rate of 0.0005%. (PDOC, p. B-7.) Proposed Condition 46 establishes a drift rate of 0.0005% and a
TDS of 5,438 mg/L in the blow down to
control PM10 emissions from the cooling tower.
However, this condition is not sufficient to limit PM10 emissions to
7.95 ton/yr because it does not limit the circulation rate to 133,378 gpm. Further, it is not enforceable as a
practical matter because it does not specify the method that would be use to
measure TDS and it does not require any demonstration that the drift rate is
actually met.
B.
The PDOC understates the PM10 increment
consumption by MEC
1.
Corrected emissions factors
The PDOC concludes that the maximum increase in 24-hour PM10 concentrations due to MEC would be 9.3 mg/m3, compared to an allowable Class II area increment of 30 mg/m3 (PDOC, Table E-8). As discussed in the preceding sections, the PDOC appears to substantially understate the PM10 emissions associated with the MEC turbines and duct burners, based on data for other projects using Westinghouse 501 turbines. The PDOC may also have underestimated the PM10 emissions associated with the cooling tower, since it relies on an unverified drift fraction. And of course the PDOC makes no allowance at all for PM10 due to ammonium nitrate formed from
ammonia slip due to use of SCR for NOx control.
After making the appropriate corrections, the District should recompute the incremental increase in maximum PM10
concentrations due to MEC.
2.
Alternative modeling approaches
The
PM emissions from the 10 cooling towers contribute significantly to the ambient
air concentrations of PM10 concentrations. The effluents have low
exit temperatures, low exit velocities and correspondingly are low in momentum
and buoyancy. When ISC3 is used to model the impact of these emissions as point
sources, as was done in the Applicant’s submission, ISC3 models these as point sources
and the Briggs plume rise equations are used.
The plumes are predicted to have significant plume rise and the ground
level concentrations are predicted accordingly. Observing that the releases often show little buoyancy, an
alternative method of modeling the emissions is to assume they emanate from a
series of volume sources based upon the dimensions of the individual cooling
towers. This method better simulates the observation that there is little plume
rise. When volume source configurations
replace the point source approximations in ISC3, the predicted concentrations
increase substantially, from a maximum value of about 8 mg/m3 on a 24-hour basis to a value of 61 mg/m3. A value of 61 mg/m3 would constitute an exceedance of the Federal PSD
increment of 30 mg/m3 for PM10.
Because of the sensitivity of the predicted concentrations to the way
that the cooling tower emissions are modeled, the District should
attempt to use alternative approaches to test whether the
cooling towers would cause an exceedance of a PSD increment.
X. THE health risks
OF MEC ARE SIGNIFICANT
The Applicant evaluated the public health impacts of the Project to comply with the District's "Toxic Risk Management Policy" ("TRMP") and to satisfy its AB2588 LORS obligation under the Warren Alquist Act. The District's policy requires the installation of Toxics Best Available Control Technology ("TBACT") if the cancer risk is greater than one in one million or if the chronic hazard index is greater than one.
The
most recent version of the Applicant's analyses are contained in Supplement C
to the AFC and in responses to data requests in the CEC proceeding, which have
been adopted in the PDOC as complying with the District TRMP. (PDOC, pp. 16-17, Appx. D.) These analyses underestimate health risks. When the errors and omissions discussed
below are corrected, the Project is found to result in significant cancer,
chronic, and acute health impacts, requiring additional mitigation and/or
denial of the Project.
A.
Cancer Impacts From The Turbine Are Significant
The
Applicant estimated incremental cancer risk (number of additional cancers per
million people exposed) using the CAPCOA Risk Assessment Guidelines.[89] (AFC, § 8.6.) Incremental cancer risk is calculated by estimating toxic
emissions, modeling these emissions to
estimate corresponding ambient concentrations,
multiplying the modeled ambient
concentration by a cancer unit risk factor, and summing over all
compounds. A cancer unit risk factor
expresses an individual’s risk of contracting cancer for a given amount of
pollutant breathed. It is expressed as
the cancer risk per amount of a pollutant in a volume of air (i.e., risk per mg/m3). Risk factors are published on the Office of
Environmental Health Hazard Assessment's ("OEHHA's") website.[90]
1.
Partial Load Operation
The
Applicant's calculations, summarized in the PDOC, suggest that the total cancer
risk is 0.2 in one million, which is less than the significance threshold of
one in one million. These calculations
assume that both turbines are operating simultaneously at full load with the
duct burners firing. However, emissions
of some toxic compounds are substantially higher during reduced loads, such as
occur during startup, shutdown, and partial load operation, than during routine
operation.
The
Gas Research Institute ("GRI") investigated the effect of load on
criteria and toxic pollutant emissions from nine gas turbines including a large
Frame 7 turbine. This study found that
emissions of benzene, toluene, formaldehyde, methane, and total nonmethane
hydrocarbons increase with load.
Emissions of formaldehyde, a carcinogen, increased dramatically, by up
to a factor of 343 when the load was reduced from 100% to 20%. For the 750 MW GE Frame 7, the formaldehyde
emission factor increased from 15 lb/1012 Btu to 7,539 lb/1012
Btu, or by a factor of 503, and the formaldehyde emissions increased from 0.11
to 16.08 tons/yr or by factor of 146, when the load was reduced from 100% to
30%. (GRI 8/96,[91]
Table S-5.) This substantial increase
in formaldehyde emissions during reduced load operation was not taken into
account in the Applicant's risk calculations.
The
maximum annual emission calculations in the PDOC are based on 520 1-hour hot
startups per year and 104 3-hour cold startups per year. (PDOC, p. B-5.) Therefore, the facility will be operating at reduced load during startups for up to 832 hours per year, or perhaps even more.[92] One or
both turbines may also operate at reduced load for additional hours during non-startup periods, which are ignored here, but which would generally
increase formaldehyde emissions beyond those claimed below. Using the Applicant's formaldehyde emission
factor of 0.11 lb/MMscf (PDOC, Table B-8) for 7,928 hour per year (8,760 hr - 832 hr = 7,928 hrs) and
an emission factor that is 146[93]
times higher for the 832 hours of partial load operation results in annual
load-weighted formaldehyde emissions that are 14.8 times higher than those evaluated
by the Applicant in the cancer risk analysis.[94] This increases the cancer risk due to
formaldehyde alone from 6.30x10-8 to 9.3x10-7.[95]
The
change in formaldehyde risk alone would increase the total cancer risk of the
Project from 0.2 in one million (0.20x10-6) assumed in the PDOC
(Table 7) to slightly over one in one
million (1.07 x10-6). Acetaldehyde is an aldehyde that is chemically
similar to formaldehyde. Assuming that
it increases in like fashion, the cancer risk from acetaldehyde alone would
increase from 1.76 x 10-8 to 1.99 x 10-7. Benzene
and possibly other toxics would also increase, further increasing cancer
risk. This exceeds the significance
threshold of one in one million and requires that TBACT be installed. At a minimum, TBACT would be either an
oxidation catalyst or SCONOx, both of which have been demonstrated to reduce
aldehydes. (Comment --.)
We note here that the PDOC condition purporting to
eliminate formaldehyde emissions to 1898 pounds per year (proposed condition 26) is no substitute for TBACT.
Compliance with proposed condition 26 is to be done by calculating formaldehyde emissions based on
“the highest emission factor determined
by any source test” of the turbines and HRSGs (proposed PDOC condition 29).
Unless the source testing is done during startup or low-load conditions it will not reveal the high formaldehyde
emission rates identified by GRI. A refinement to condition 29 requiring source
testing at low load and/or start-up conditions should be an additional MEC
condition, in addition to requiring TBACT for aldehydes.
2.
Dry Low NOx Combustors
The
Applicants used CARB's CATEF database to estimate toxic emissions of all
chemicals except acrolein (which is discussed in Comment --). We suspect that the combustors on the
turbines used to develop these emission factors were based on the diffusion-flame
principle and were not equipped with pre-mixed, dry low NOx combustors, which
are now widely used to control NOx. The
CATEF database is based on measurements that were made between 1988 and 1992,
when steam injection was predominately used to control NOx rather than
pre-mixed combustors.
The
GRI study included in Exhibit 44indicates
that pre-mixed low NOx combustors increase emissions of formaldehyde by about a
factor of 6.6 during full load and by a
factor of 11.4 during reduced load,
compared to the same turbine without a low-NOx combuster. (Ex. 44
Table S-5, p. 8.) Benzene
emissions are almost 40 times greater during
reduced load operation for a turbine with low-NOx combustors as for the
same kind of turbine without them, and are over twice as great during full-load
operation when a low0NOx combustor is used (ex. 44, Table S-5, p.8)
The
Project proposes to use a dry low NOx combustor to control NOx. Therefore adjustment factors have been
included in Table 8 to account for increased emission associated with turbines
using low-NOx combustors.
3.
Other Carcinogens
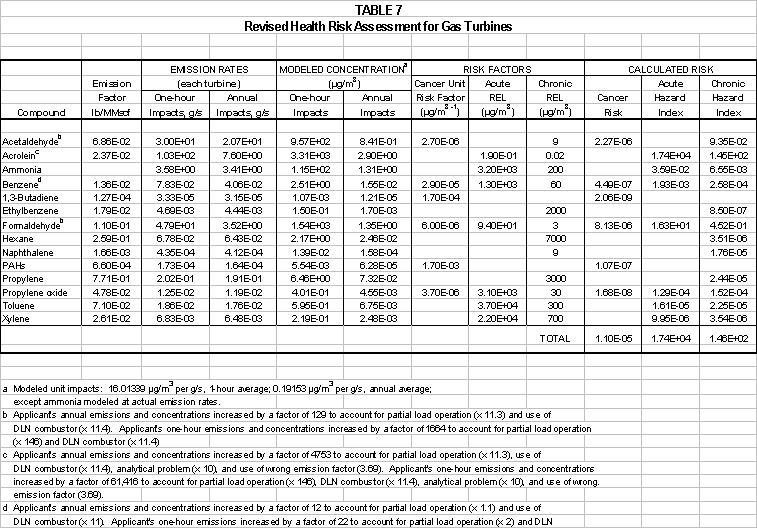
The Applicant's analysis only considers five
carcinogens: acetaldehyde, benzene, 1,3-butadiene, formaldehyde, and PAHs. However, dioxins, furans, hexavalent
chromium, lead, and nickel are
present in the emissions from Frame 7
machines. (ESI 6/30/92.[96]) We did not include them in our analysis
because cancer risks exceed significance thresholds without them. However, the Applicant should be required to
revise its risk calculations to include these additional substances, because
they may make a significant contribution when considering controls to reduce
risks from the volatile organic compounds discussed above.
B.
Cancer And Noncancer Impacts From Emergency Diesel
Engines Are Significant
The
CEC and the District required the Applicant to evaluate the health impacts of
diesel exhaust particulates from the standby fire pump diesel engine in
anticipation of pending amendments to District Regulation 2, Rules 1 and
2. (PDOC, p. D-1.) The firewater pump is a 300-hp Cummins engine that would be
tested for about 30 minutes each week, up to 100 hours per year. The Applicant's risk calculations are
included in its response to CEC data request PH-2 and indicate that the total
cancer risk is 0.89 in one million, which is less than the significance
threshold of one in one million. The
Applicant's calculations also indicate that the 1-hour concentration of diesel
exhaust is 12 ug/m3, which is less than the acute reference exposure
level of 50 ug/m3 and the annual average concentration of 2.98x10-3
is less than the chronic reference exposure level of 5 ug/m3. (Metcalf Response to CEC Data Request PH-3,
4/7/00, p. 5.) Therefore, according to
the Applicant, diesel exhaust from the fire pump engine would not cause
significant health impacts. (PDOC, pp.
16, D-2.) However, there are two
problems with the Applicant's analysis, which substantially underestimate
cancer risk.
First,
the Applicant performed its calculations for the nearest residence. Health risks are usually calculated at the
point of maximum impact. (CAPCOA
11/93.) This is particularly critical
in a rapidly growing area where land use is
changing, such as the Project site.
Therefore, we remodeled the fire pump using the Applicant's emissions
and stack parameters to determine the point of maximum impact and corresponding
concentrations of diesel exhaust. This
analysis shows that the 1-hour concentration of diesel exhaust increases from
12 ug/m3 at the nearest residence to 52.5 ug/m3 at the
point of maximum impact (UTM 611400, 4119720).
Similarly, the annual
concentration increases from 2.98x10-3 ug/m3 at
the nearest residence to 9.29x10-3 ug/m3 at the point of
maximum impact (UTM 611400, 4119720).
Assuming
the point of maximum impact is developed for commercial use in the future,
which cannot be precluded given growth in the area, the cancer risk increases from 0.89x10-6
claimed by the Applicant to 2.79x10-6,[97]
which exceeds the significance threshold of one in one million. Similarly, the maximum 1-hour concentration
of 52.5 ug/m3 exceeds the acute REL of 50 ug/m3. The corresponding hazard index (52.5/50 =
1.05) exceeds the significance threshold of one. Therefore, acute and cancer impacts from the firewater pump are significant, requiring mitigation. Installing Engelhard or equivalent
soot filters can control Diesel exhaust from firewater pumps.
Second,
the Applicant modeled the subject pump assuming a 35 ft high vertical
stack. The pump is located within a
24-foot high building. (AFC, Figs.
2.2-1 and 2.2-2.) Typically, pump
stacks that are vented out of a building are run up the side of the building to
about 5 feet above roof level.
Therefore, the 35-ft high stack modeled by the Applicant is on the high
side, underestimating ambient concentrations.
Further, the exits of these types of stacks are conventionally outfitted
with a rain hat or a gooseneck to keep
out rain. Emissions from rain-hooded
and gooseneck vents would be deflected
downward towards the roof. Therefore,
they have no vertical buoyancy and are normally modeled with an exit velocity
of zero or as a fugitive volume source.
We
remodeled the firewater pump with an
exit velocity of zero. This increased
the maximum annual concentration to 1.23x10-2 ug/m3 and the corresponding
cancer risk to 3.68x10-6.
Therefore,
the Applicant has substantially underestimated health impacts of diesel exhaust
from the emergency pumps. Diesel
exhaust particulate concentrations exceed significance thresholds, which require that the District require controls.
C.
Noncancer Health Impacts From Turbines Are
Significant
The
Applicant estimated Noncancer health
impacts using the hazard index approach.
A hazard index is the ratio of the ambient concentration, estimated by
modeling, to the reference exposure level or REL, which is the concentration
deemed by OEHHA as being safe.[98] The total hazard index is the sum of the
indices for individual compounds. If
the chronic hazard index exceeds one, impacts are significant and TBACT is
required. The District does not
evaluate the acute hazard index, but the Energy Commission typically considers
an acute hazard index greater than one as significant. The Applicant estimated a chronic hazard
index of 0.06. (AFC, p. 3-4; PDOC,
Table D-2.) As discussed above, this
index is underestimated because the Applicant's calculations do not take into account-increased emissions during partial load operation and the
use of a DLN combustor. In addition,
the Applicant made a questionable adjustment to the acrolein emission factor.
1.
Acrolein Emission Factor
The
Applicant originally estimated acrolein emissions using the CARB emission
factor of 2.37x10-2 pounds per million standard cubic feet of gas
combusted ("lb/MMscf").
However, these original calculations were based on outdated acute
reference exposure levels ("acute RELs"). Thus, the Energy Commission directed the Applicant to revise
their health risk assessment to use current acute RELs. (CEC Data Request 63, 8/23/99, p. 62.) This increased the acute hazard index from
0.16 (AFC, Table 8.6-6) to 1.05,[99]
exceeding the significance level of one.
Therefore, the Applicant apparently reduced the acrolein emission factor
from 2.37x10-2 to 6.43x10-3 to reduce acute health impacts below the
significance level of one. (AFC,
Supplement C, Table 8.1A-3.) This
adjustment is wholly unsupported.
Both the Energy Commission Staff (CEC Data Request PH-1, April 7, 2000) and the District (Jang 3/2/00[100]) requested a clarification of this change. The Applicant explained that the original emission factor was based on four source tests, only one of which was conducted on a Frame 7 machine. Therefore, the Applicant used the single source test on the Frame 7, which lowered the acrolein emission factor from 2.37x10-2 to 6.43x10-3 lb/MMscf. This is not justified as explained below.
Acrolein
is the most toxic compound emitted by the gas turbines. It is a double-bonded aldehyde which causes
eye, nose and throat irritation. It has
the lowest acute and chronic reference exposure level among all of the
substances emitted by the turbine.
Therefore, very small concentrations of acrolein, much smaller than any
other compound emitted by the Project, will result in significant health
impacts.
The
Applicant's risk assessment relied on a revision to the acrolein emission factors
reported in CARB's CATEF database.
However, the acrolein emission factor in this database was based on
source tests in which acrolein was measured by CARB Method 430. (CARB 4/96.[101]) CARB has recently published an advisory that
states: "any data or results, based on the use of M430 to determine
acrolein...are suspect and should be flagged as nonquantitative wherever they
appear." (CARB 4/28/00.[102]) This method has been validated for only
formaldehyde and acetaldehyde and substantially underestimates acrolein
concentrations.
CARB
Method 430 measures acrolein by reacting it with 2,4-dinitrophenylhydrazine
("DNPH") acidified with hydrochloric acid, which converts the
acrolein into its hydrazone derivative.
The hydrazone derivative is then analyzed by high performance liquid
chromatography. However, the hydrazone
derivatives of double-bonded aldehydes such as acrolein are not stable under
acidic conditions and rapidly degrade.
The acrolein hydrazone derivative begins to degrade almost
immediately. After 30 minutes, 60% is degraded, after 1 hour, 70% is
degraded, and after 48 hours, 93% is degraded. (Exhibit --: Freeman 1993.[103]) Typically, it takes four hours to collect a
sample. One to two weeks typically
elapse between sample collection and analysis.
Therefore, well over 90% of the acrolein is lost before the sample is
analyzed.
The
Applicant justifies using the Frame 7 emission factor "[b]ecause of the
differences in combustion and control technology between the frame turbines and
other turbines tested, there is no reason to believe that the test results from
the other turbines are more representative than the test results from the frame
turbine." (Metcalf Response to
Staff Data Request PH-1, 4/7/00, pp. 1-2.)
However, differences in the elapsed time between sample collection and
extraction among the four source tests creates far more variability among
acrolein emissions than turbine type.
Thus, it is impossible, given this situation, to conclude that any one
of the four measurements is any more representative of Frame 7 machines than
any other. In fact, it is a fair bet
than none are representative and all are underestimates. Therefore, none of the data should have been
discarded. To address these analytical
problems, we multiplied the acrolein emission factor by ten to adjust for the
degradation that occurs between sample collection and extraction. We believe that this is conservative and
underestimates actual acrolein emissions.
Using
the average CATEF emission factor (2.37x10-2 lb/MMscf) as required
by CARB[104] and
increasing it by a factor of ten increases the acute hazard index from 0.33
reported in the PDOC to 1.7, which is significant. Likewise, these same two changes (CATEF data instead of the Applicant’s unilateral
adjustment of it; factor of ten increase to reflect CARB 430 under
measurement of acrolein) increase the chronic hazard index from 0.06
reported in the PDOC to 0.93.
In addition to using the wrong
emission factor and failing to adjust it to account for acrolein degradation,
the Applicant also failed to consider startup and shutdown conditions and the
use of DLN combustors, as discussed above.
Each of these factors further increases the chronic and acute hazard
indices. Incorporating all of these
factors, as explained in Table 8,
increases the acrolein hazard index values in Table 7, which are far
above the permissible level of 2. Even if
acrolein emissions are overestimated by factors of ten to one
hundred, these hazard indices would still exceed the significance threshold of
one by a large margin, requiring the use of TBACT. Put another way, if
any of several acrolein emissions adjustment factors listed in Table 8 are
appropriate, then acrolein emission adjustment factors listed in Table 8 are
appropriate, then acrolein will have an acute hazard index greater than one.
2. The Acute Hazard Index for Fermaldehyde Also Exceeds One
Acrolein is not the only compound
shown in Table 7 with a revised acute hazard index greater than one. The index
for formaldehyde also exceeds one, providing yet another reason (besides its
cancer risk) to require BACT for formaldehyde.
XI.
The PDOC’s analysis of visibility impacts is
inadequate
The
PDOC fails to satisfy the requirements of federal regulations regarding the
protection of visibility. The PDOC
states, with no factual or analytical support, that "....the proposed
project will not cause any impairment of visibility at Pinnacles National
Monument..." However, the
District's own regulations, at Rule 2-2-309,
state that a project must be denied "where it has been demonstrated
by the Federal Land Manager that the permit would authorize emissions which
would have an adverse impact on the air-quality-related values (including
visibility) of a Class I area..."
Nowhere in the District's PDOC is there any mention of input from the
responsible Federal Land Manager, whose substantive input on this question is indispensable in order to adequately address the question of the
potential impacts of a proposed project on air-quality-related values in a
Class I area. EPA has designated
the Pinnacles National Monument as a mandatory
Class I area where visibility is an important value. See 40 CFR section 81.405.
Pinnacles is 60 miles from, and is directly downwind from, the
proposed Metcalf Energy Center. Under
EPA's regulations, the agency reviewing the permit application, in this case,
the District, must provide notification to the responsible Federal land manager
responsible for a Class 1 area that might potentially be impacted by a project
under review and that the reviewing agency must consider any analysis performed
by the Federal land manager on this issue.
See 40 CFR section 51.27(d). At
no point in the PDOC is there any indication that the District even consulted
the Federal land manager for Pinnacles, much less than that any such comments
from the Federal land manager were considered by the District as part of its
air quality impacts analysis. The
District's visibility analysis is therefore fatally flawed.
Furthermore,
under EPA's rules, any person has the right, in connection with an application
for a permit to construct a major stationary source (and the Metcalf project
certainly qualifies under EPA's definition as a major
stationary source, both for NOx and PM10) to request that the Administrator of
EPA take responsibility from the state for conducting the required review of a
proposed source's impact on visibility in any Federal Class I area, and that if
so requested, the Administrator shall take such responsibility
and conduct such review. See 40 CFR
section 52.27(c). Given the proximity
of Pinnacles to the proposed project location and the fact the proposed project
location is directly upwind of Pinnacles, the District's apparent total failure
to take seriously its responsibility to consult with the responsible Federal
land manager for Pinnacles leaves a gaping hole in the required analysis for
this project. In light of this failure,
the Energy Commission staff should and must assert its right under section
52.27(c) and bring EPA in to conduct the very important consultation on
visibility impacts of the Metcalf project with the Federal land manager for
Pinnacles that the District has apparently failed to perform.
XII.
OTHER ISSUES
A.
EMERGENCY IC ENGINES NOT PROPERLY PERMITTED
The Project includes two
emergency internal combustion (“IC”) engines, a 300-hp, diesel-fired fired
Cummins engine used in a fire-water pump and a 6.44 MMBtu/hr, natural gas-fired
Caterpillar engine used in an emergency generator. These two emergency engines are exempt from District rules
pursuant to Regulation 1-110.2. (PDOC,
p. 2.) However, they are not exempt
under federal PSD regulations at 40 CFR 52.21.
The Draft Permit does not even mention these two engines, let alone
establish BACT and enforceable permits limits as it must to comply with federal
law.
The federal PSD program requires
that BACT be applied to each emission unit at a major stationary source that
would have the potential to emit in significant amounts. (40 CFR § 52.21.) The EPA as applying to “each individual new or modified affected
emissions unit and pollutant emitting activity at which a net emissions
increase would occur has interpreted the applicability of BACT. Consequently, the BACT determination must
separately address, for each regulated pollutant with a significant emissions
increase at the source, air pollution controls for each emissions unit or
pollutant emitting activity subject to review." (NSR Manual, p. B.4 (emphasis added).)
The PDOC acknowledges that
Metcalf's emissions exceed the significance thresholds in 40 CFR § 52.21(b)(23)
for NOx and CO. (PDOC, p.
7.) We believe the thresholds are also
exceeded for ozone and PM10, as discussed in Comment --. Thus, Metcalf is a major stationary source
and BACT is required for each emission unit at Metcalf, including the two
emergency IC engines. The District must
perform a formal, top-down BACT analysis for these two engines, establish BACT
emission limits,[105]
and impose enforceable permit limits which limit both emissions and hours of
operation. The resulting analyses and
proposed permit limits must be circulated for public review. (NSR Manual, p. B.56.)
I.MISCELLANEOUS PERMIT condition ISSUES
Cooling Tower Emissions Not
Properly Limited
The cooling tower would emit 7.95 ton/yr of PM10,
calculated assuming a cooling tower circulation rate of 133,378 gpm, a maximum
total dissolved solids ("TDS") of 5,438 mg/L, and a drift rate of
0.0005%. (PDOC, p. B-7.) Proposed Condition 46 establishes a drift
rate of 0.0005% and a TDS of 5,438 mg/L in the blowdown to control PM10
emissions from the cooling tower.
However, this condition is not sufficient to limit PM10 emissions to
7.95 ton/yr because it does not limit the circulation rate to 133,378 gpm. Further, it is not enforceable as a practical
matter because it does not specify the method that would be use to measure TDS
and it does not require any demonstration that the drift rate is actually met.
Draft Condition 47 requires that the owner/operator
perform a "visual inspection" of the drift eliminators at least once
per year and make repairs as necessary.
It further requires that the vendor inspect the drift eliminators and
certify that they are properly installed.
However, nothing in the Draft Permit actually requires a demonstration
that the drift eliminators meet 0.0005%.
Without this demonstrate, the 0.0005% drift fraction is a hollow
promise.
Condition 47 should be expanded to require
submittal of a vendor's guarantee at least 30 days prior to commencement of
construction. Further, it requires no
demonstration that the 0.0005% drift rate would actually be met. It generally is not practical to source test
a cooling tower. Normally, compliance
with drift permit conditions is demonstrated through an annual performance test
to verify the operating efficiency of the drift eliminators. (See permits in Exhibit 14.) This test should be performed by a licensed
Cooling Tower Institute drift testing firm and should, at a minimum, sample two
cells with a minimum of 3 runs per cell.
B.
Source
Testing Protocol Is Not Adequate
According to the NSR Manual,
"[t]he construction permit should state how compliance with each
limitation will be determined, and include, but not be limited to, the test
method(s) approved for demonstrating compliance. These permit compliance conditions must be very clear and
enforceable as a practical matter (see Appendix C). The conditions must specify:
· when and what tests should be performed;
· under what conditions tests should be performed;
· the frequency of testing;
· the responsibility for performing the test;
· that the source be constructed to accommodate such testing;
· procedures for establishing exact testing protocol; and
· requirements for regulatory personnel to witness the testing. (Id., p. H.6.)
Source testing requirements in Conditions 12, 30, and 31 do not specify the test methods, the conditions under which the tests would be performed (e.g., startup, shutdown, 50% load, duct burners on or off, steam injection for power augmentation), the responsibility for performing the test, procedures for establishing an exact testing protocol, and a requirement for regulatory personnel to witness the testing. Instead, the Draft Permit specifies "District-approved" methods (which are not identified specifically) and allows the development and approval of source test procedures prior to conducting any tests by the District, outside of the public view. In fact, Condition 39 allows the owner to contact BAAQMD "regarding requirements for the continuous monitors, sampling ports, platforms, and source tests required by conditions 32, 33, and 35." This information should all be in the Draft Permit and thus subject to public review.
Therefore, the Permit does not
establish the conditions required to determine compliance, but rather leaves
the establishment of such provisions to the future discretion of the District
in approving a source test protocol.
There is no assurance that the establishment of a future protocol would
be subject to the public notice and review requirements of 40 CFR 52.21 &
124. Therefore, relying on a future
source test protocol as advocated here is clearly erroneous as it allows for
specification of the terms of the PSD permit outside of the PSD permitting
process.
C.
A
CEMs Should Be Used To Assure Compliance With The SO2 Limit
The EPA specifically requires that “[p]arameters which must be monitored continuously or continually are those used by inspectors to determine compliance on a real-time basis and by source personnel to maintain process operations in compliance with source emissions limits.” (NSR Manual, p. H.7.) The Draft Permit only requires that SO2 be measured annually in a source test and otherwise, that fuel sulfur be analyzed monthly. (PDOC, p. 34, Condition 45.) It is well known that fuel sulfur content can be quite variable, and spikes are common. Therefore, this condition is not adequate to assure continuous compliance. The Permit should be modified to require SO2 CEMs.
D.
Use of 1 year of offsite meteorological data is
inconsistent with Federal EPA guidance.
The air dispersion modeling performed to calculate the PSD increment consumption and air quality impact for comparison to the NAAQS and California AAQS is based upon the use of only one year of meteorological data obtained at a location (IBM site) nearly 5 kilometers from the proposed Metcalf Energy Center site. As discussed below, EPA will allow the use of a minimum of one year of data if the data is site-specific and representative. Otherwise, EPA requires that five years of “ adequately representative” data be used. The reason for this difference in the required duration of measurements is that EPA recognizes the importance of site-specific meteorological data to the validity of model predictions and wanted to encourage sources to set up and collect on-site meteorological data collection systems for input to dispersion modeling for regulatory applications. To mandate a full five-year duration on-site program was deemed to cause an unacceptably long delay to the permitting process.
The use of meteorological data from the IBM site, however, does not pass the site-specific test as it is located in a very different and wider part of the Santa Clara Valley. As such, the meteorological data is not representative of the meteorological conditions affecting plume dispersion of effluent plumes at the proposed complex terrain site.
EPA’s current guidance on these issues are in two related documents: Appendix W to Part 51-Guideline on Air Quality Models (1999 Edition) and a related, referenced document EPA-450/4-87-013: On-Site Meteorological Program Guidance For Regulatory Modeling Applications. Appendix W Section 9.3.3.1 Discussion (under the heading of ‘Site-Specific Data’) states that “Spatial or geographic representative ness is best achieved by collection of all of the needed model input data at the actual location of the source(s)”. Section 1.2 of the ‘On-Site’ guidance document provides the following definition: “On-site refers to the collection of data at the actual site of a source that are representative, in a spatial and temporal sense, of the dispersion conditions for the source.” Both of the above quotations are in the context of any terrain situation (e.g. flat or complex). For complex terrain settings (The proposed MEC site is clearly complex terrain with Tulare Hill to the west 150 feet above stack top and peaks 3 km to the east over 1100 feet above stack top.), the guidance is even more specific. Appendix W Section 9.3.3.2 (h) states that” For refined modeling applications in complex terrain, multiple level (typically three or more) measurements of wind speed and direction and turbulence (wind fluctuation statistics) are required. Such measurements should be obtained up to the representative plume heights of interest…” Similarly, the ‘On-site’ guidance Section 3.2 Complex Terrain Sites states “The ideal siting solution in complex terrain involves siting a tower between the source in question and the terrain obstacle of concern. The tower should be tall enough to produce measurements at the level of the plume, and should provide measurements of all variables at several levels.” Clearly, data from the 10-meter high anemometer at the IBM site does not meet the above proximity and representative ness criteria. It certainly does not provide measurements at the level of the expected plumes of concern.
We note that EPA, on April 21, 2000, proposed revisions to Appendix W, which, if adopted, would change the wording of some of the above. The revised Appendix W also references a new document: Site-Specific Meteorological Monitoring Guidance for Regulatory Applications (1999) (EPA –454/R-99-005). We discuss relevant word changes in these documents below. We do not believe that they alter the conclusion that the meteorological data utilized in the PDOC does not meet either present or proposed changes to their guidance criteria. The April 21, 2000 proposed revision to Appendix W to Part 51-Guideline on Air Quality Models pertaining to the length of Meteorological records states in Section 8.3.1.2 Recommendations (a) that “ Five years of representative meteorological data should be used when estimating concentrations with an air quality model. Consecutive years from the most recent, readily available 5-year period are preferred. The meteorological data should be adequately representative, and may be site specific or from a nearby NWS station.” Section (b) goes on to state that “The use of 5 years of NWS meteorological data or at least 1 year of site-specific data is required. If one year or more of, up to five years, of site-specific data is available, these data are preferred for use in air quality analyses.” Section 8.3.3.2 clarifies the meaning of ‘site-specific’: adding “As a minimum, site-specific measurements of ambient air temperature, transport wind speed and direction, and the variables necessary to estimate atmospheric dispersion should be available in meteorological data sets to be used in modeling. Care should be taken to ensure that meteorological instruments are located to provide representative characterization of pollutant transport between sources and receptors of interest.” We note that whereas in EPA’s current Appendix W, Section 9.3.3.1 defines the needed model input data “ at the actual site of the source(s)”, the proposed revision to the same section (renumbered to be 8.3.3.1) uses the phrase “in close proximity to the actual site of the source(s)” and goes on to state that “collection of meteorological data on property does not of itself guarantee adequate representative ness.” As discussed elsewhere, in the context of the needs for the present application, the IBM site is neither “on-site” nor “in close proximity”. In EPA’s new Meteorological Monitoring Guidance for Regulatory Modeling Applications (February, 2000), in Section 3.3 concerned with sources in complex or mountainous terrain, EPA discusses how ”…measurements should be made at multiple levels in order to ensure data used for modeling are representative of conditions at plume level. The ideal arrangement in complex terrain involves siting a tall tower between the source and the terrain feature of concern.” The messages are the same in both the present and potentially revised guidance: One needs to have very representative data for model inputs. The proximity issue for an anemometer in complex terrain focuses, in addition, also on the elevation of the measurements with respect to the range of plume heights of concern.
The rationale behind this guidance is the importance of measuring the wind speeds and directions that would affect plumes that would travel toward areas of high terrain, or in the case of terrain induced downwash, from high terrain areas toward the stack locations. The predicted concentrations are very sensitive to the very local meteorology in complex terrain settings. Tulare Hill is a significant local obstacle to the prevailing up and down valley flows expected in the Santa Clara Valley. It rises over 300 feet above the stack bases and about 150 feet above the tallest of the turbine stacks. The airflow between the MEC site and the hill will clearly be affected by the presence of Tulare Hill. The alterations of the flow patterns will, in turn, affect the dispersion of emissions from all the sources at the MEC site. In the narrow section of the Santa Clara Valley between closest to the proposed MEC, one would expect locally higher wind speeds during periods of general up or down valley flows. To the extent that these higher wind speeds affect the plumes from the MEC, we would expect to see a higher frequency of building induced downwash events affecting the air quality in the immediate vicinity of the site. Terrain induced downwash occurring when winds from the north and west flow over portions of Tulare Hill increase ground level concentrations to the east and south of the site. We note that the ambient air quality at the proposed development of the Cisco Systems Campus and associated day care and other populated areas is likely to be routinely adversely affected by these downwash conditions. It is important that winds at stack height be measured and that a longer database be developed. It is very important in complex terrain settings that the anemometer height be high enough to characterize the winds at stack top and above stack top. As noted above, EPA recommends that measurements be taken at multiple levels to achieve this requirement. The anemometer at the IBM site is at 10 meters above the surface. It cannot be relied upon to estimate winds at the 44-meter heights of the turbine exhausts or used to infer winds at the final rise heights. This is a clear case where a site-specific meteorological monitoring program that collects a year or more of data is required. The use of meteorological data from a site 5 km away cannot capture the site-specific flows of concern to the neighbors of the proposed MEC.
C. The use of
‘Rural’ dispersion coefficients needs to be re-examined for this
application
The applicant has used the Auer
method for determining that rural dispersion coefficients are appropriate for
the modeling effort based upon present land use in a circle surrounding the
proposed site. The ISC3 model forces a
single choice for the use of Urban vs. Rural coefficients. Urban coefficients reflect greater surface
roughness and result in more vigorous vertical mixing of pollutants emitted
into the lower atmosphere. It is likely that a determination of urban would
result in higher predicted concentrations in populated areas to the northwest
and southeast of the site because elevated plumes would rapidly mix downward to
the ground. The Auer method simply
requires an examination of land use within a 3-kilometer circle around the
site. The determination of rural by the applicant is largely associated with the
large areas of undeveloped land to the north east side of Route 101 and the
undeveloped land of Tulare Hill. Winds flowing from the northeast and east
across the areas to the northeast of Route 101 and toward the plant are
relatively rare and the Auer method unfairly weights them for this setting.
Although Tulare Hill has a relatively smooth surface, the hill itself causes
increased turbulence in winds flowing from the north and northwest (also known
as terrain-induced downwash) and in reality should not be contributing to the
support of a rural dispersion coefficient determination.
In an area dominated by dominant and specific prevailing winds from two directions, a more realistic determination would weight the land uses by the percentage of the time that the winds transporting pollutants from the plant are along those wind directions. Those directions coincide with the directions of high population density. Note that the planned campus development in the area to the south of the proposed plant location would not only change the surface roughness by the construction of buildings but also greatly increase the population density of potentially exposed people. It is important to also observe that the US EPA is aware of the deficiencies of ISC3 and the Auer method in applications such as this one. EPA has recently proposed to replace the ISC3 model with an improved model, AERMOD. AERMOD’s primary difference from ISC3 is the allowed and encouraged use of more site specific and detailed meteorological data as input to the model. For the present application, AERMOD will allow the use of direction specific roughness coefficients, which would in effect increase the vertical mixing of pollutants as the plumes passed over built-up or rougher terrain areas. The determination of direction specific roughness parameters would essentially eliminate the use of the Auer method and should yield more realistic predictions of air quality concentrations. With the mix of higher elevation and lower elevation sources at the MEC, the effects of an urban dispersion coefficient determination should be evaluated. Alternatively, the AERMOD model should be run after an assessment of present and future roughness lengths has been made.
D. Potential for simultaneous turbine start-up
PDOC proposed licensing condition 22 requires the two gas turbines at MEC not to be in start-up
simultaneously. The PDOC indicates a 3-hour duration for cold start-ups (PDOC, p.
B-5), and in Table B-13 assumes one gas turbine in start-up at the
beginning of a 24-hour period, with the
other starting up “at beginning of third hour of 24 hour period.” Were the situation in Table B-13 to occur in real
life, MEC would be in violation of proposed condition 22.
How likely is it that simultaneous turbine start-up
could occur? Calpine/Bechtel Enterprises will have substantial
financial incentives to start both
turbines at once. The two turbines will be basically identical, with identical
operating costs, and they will be
operated as merchant facilities in response to market prices. So whenever market conditions justify operating one turbine, they
will justify operating both, and whenever market prices justify shutting down a
turbine they will justify shutting down both. Compliance with condition 22
therefore will require Calpine/Bechtel to either start up the first turbine
before it is cost-effective to do so, or
delay starting up the second one until after it is cost-effective to do so.
California energy prices can change by many dollars
per Mwh from one hour to another. For a 300 Mw turbine/HRSG train as proposed
at Metcalf, that means the foregone profits from a delayed start-up of one
turbine could easily run to thousands of dollars per hour. With the Applicant
proposing up to 312 starts per year per turbine, the financial cost of
complying with condition 22 will be substantial.
Nevertheless, compliance with condition 22 is
essential. Many of the MEC emissions will be higher during startup than during
normal operations, and allowing both units to be in startup at once would
produce substantially greater one-hour
emissions than the PDOC has assumed. Therefore the PDOC conditions must be
modified to include a monitoring and enforcement provision for condition 22.
The proposal made above that the CEMS for MEC be required to be operable and
accurate during startups are one way that any violation of condition 22 would
be detectable. In addition, we propose that the Applicant be required to keep a
real-time log of unit output (e.g., the
same sort of data it would report to the California Independent System Operator (CAISO) in the normal course of business as an
ISO-interconnected facility) showing unit output for each turbine in intervals
of no more than 10 minutes (the ISO already operates using 10-minute
data). This data should then be provided
to the District at least monthly to verify that at no time were both turbine
units in startup (ramping up their output) or shutdown (ramping down their
output).
E.
Verification of ERCs
The PDOC asserts that Calpine/Bechtel have identified valid emission reduction credits (ERCs) of 356 tons/year of POC from banking certificate #625 belonging to Quebecor, Inc. of San Jose (PDOC, Table 4, p. 15). ERCs are required to be federally enforceable (PDOC, p. 13). Nowhere in the PDOC is there any analysis of the Quebecor ERCs to verify their validity. In particular, the PDOC should be amended to include a discussion of whether the Quebecor ERCs were properly issued and quantified, and whether an analysis of BARCT would reduce the size of the Quebecor ERCs. Since the Quebecor ERC certificate represents over 10 percent of all the POC ERC’s in the BAAQMD bank, and is over twice as large as the next largest certificate in the bank, it is particularly important that the BAAQMD provide a close and public analysis of its size and validity.